International Journal of Clinical and Medical Research | Year 2025 | Volume 2 | Issue 2 | Pages 1-22
Ethnopharmacology, Phytochemistry, and Biological Properties of Medicinally Important Plants of Bignoniaceae Family: A Comprehensive Review
Malik Saadullah1, Maryam Farrukh 2*1,2,3,4Department of Pharmaceutical Chemistry, Faculty of Pharmaceutical Sciences, Govt. College University, Faisalabad-38000 Pakistan
View PDF Download XML Download DOI XML DOI: 10.66590/ijcmr2025020201
Abstract
Aim of the study: The family Bignoniaceae is also known as the "trumpet vine family". Majority of members of this family scattered throughout tropical America. Traditionally, plants of this family were used to treat various disorders, like neuralgia and varicose veins, diabetes, syphilis, ulcers, abscesses, rheumatism, gonorrhoea, and wounds. The biological characteristics, phytochemistry, and chemotaxonomic classification of the phytochemicals of several species of the family Bignoniaceae are highlighted in this paper. Materials and methods: The relevant information on the family Bignoniaceae was obtained from scientific databases (Google Scholar, ACS Publications, Wiley Online Library, Science Direct, PubMed). Information was also gathered from books and online databases. The literature cited in this review dates from 1971 to June 2022. Result: About 133 compounds have been isolated from various species of the family Bignoniaceae, including anthraquinones, sterols, terpenoids, benzoquinone, naphthoquinones, phthalide derivative, furanonaphthoquinones, phenolic acid and their derivatives, fatty acids, flavonoids, carotenoids, glycoside, dihydroisocoumarins, lignans, alkaloids, and sugars. Structures of these components are shown in tabular form. In biological studies, the crude extracts of the various species of the family Bignoniaceae show many biological properties, like antibacterial, antiviral, antifungal, antidiarrheal, anticancer, antioxidant, anticonvulsant, antidiabetic, antiplasmodial, analgesic, anti-inflammatory, antimalarial, anti-trypanosomal, antispasmodic, wound healing activity, cardioprotective, and larvicidal activity. The biological properties are also shown in tabular form.
INTRODUCTION
According to a WHO report, the majority of traditional therapies use plant extracts and their active ingredients because they are safe and effective [1]. Over 80% of people worldwide rely on natural resources (especially plants) to treat their illnesses, either because of drug-resistant conditions or the negative side effects of modern medications [2]. Some justifications for using medicinal plants include their affordability, accessibility, and the perception that they are safer than manufactured medications [3]. The majority of herbal products are complex and vary in character, whether they are made from a single or a combination of botanical substances [4].
The existence of numerous secondary metabolites in medicinal plants, such as alkaloids, flavonoids, phenols, saponins, and sterols, may contribute to their therapeutic effects [5]. Many years ago, traditional medicinal herbs were used to treat a variety of diseases [6]. The herbs have anti-depressant, anti-asthmatic, post-coital antifertility activity, spermicidal activity, anti-parasitic activity, hepatoprotective, hypoglycaemic activity, anti-inflammatory, bronchoprotective, antiallergic, and nephroprotective properties [7].
A family of trees, shrubs, lianas, and sporadically herbaceous species is known as the Bignoniaceae [4]. It is a large family that is also known as the "trumpet vine family that is also known as the "trumpet vine family" or "trumpet creeper family" [8]. With more than 100 genera and around 800 species, the Bignoniaceae family is largely tropical. Figure 1 is showing few plants of Bignoniaceae Family [9]. Majority of its members scattered throughout tropical America [10].
The Bignoniaceae family includes important ornamental plants with gigantic, striking flowers [4]. The family is distinguished by the wide variety of vibrant, beautiful flowers they produce [8]. Alkaloids, flavones, polyphenols, naphthoquinones, tannins, triterpenes, seed oils, and iridoid glucosides are the main chemical constituents that are recognised in this family [11]. It consists of species which have been utilised traditionally for a variety of conditions [12] like fungus infections [13], snakebites, skin conditions, gynaecological conditions, cancer, cholera, gastrointestinal problems, respiratory issues, urinary problems, hepatic conditions, epilepsy, sexually transmitted diseases, pain, malaria, and heart conditions [14].

Figure 1: The plants of Bignoniaceae Family (a), Newbouldia laevis (P.Beauv.) Seem. (b), Campsis grandiflora K.Schum. (c), Zeyheria montana Mart. (d), Kigelia africana (Lam.) Benth. (e), Pyrostegia venusta Miers (f) Tabebuia impetiginosa Standl
Traditional Uses
An antinociceptive and anti-inflammatory action has been seen in extracts of the pods, leaves, or seeds of Catalpa bignonioides Walter [15]. The plant Jacaranda mimosifolia D. Don is used to treat hepatitis. The bark, flowers, and leaves of Jacaranda mimosifolia are used in conventional medicine to treat neuralgia and varicose veins [16]. Tabebuia impetiginosa Standl. contains antinociceptive, antibiotic, anti-edematogenic, and antidepressant properties [17]. Diabetes can be treated with the Tecoma stans (L.) Griseb. plant [18]. Backaches, syphilis, ulcers, abscesses, rheumatism, and wounds are all treated with Kigelia pinnata. (Jacq.) DC. [19]. The flowers of Spathodea campanulata Buch.-Ham. ex DC. are used to treat ulcers in Laos, Cambodia, and Vietnam. [16]. The leaves and bark of Markhamia stipulata Seem. ex K.Schum. are used both internally for analgesic effects and externally to treat skin disorders in conventional Thai treatment [20]. Leaves of Fernandoa adenophylla (Wall. ex G.Don) Steenis are used as an external therapy for skin conditions in traditional Thai medicine [21]. White skin patches (leukoderma, vitiligo) can be treated with the help of flowers and leaves of Fernandoa adenophylla [22]. Brazilian folk medicine uses Jacaranda cuspidifolia Mart. for pectoral and vulnerable treatments, as well as for the treatment of syphilis, gonorrhoea, and skin ulcers [23].
Phyto-Constituents
Phytochemicals, which have been demonstrated to help prevent, treat, or relieve a variety of health issues, are abundant in plants and herbs [24]. Several investigations on various plant species have been conducted recently in an effort to find chemicals that could be useful for various medical uses [25].
Compounds 1–133 were isolated from medicinally important genra of Bignoniaceae (Table 1). Commonly, Naphthoquinones are the major constituents of this family. Many terpenoids, anthraquinones, benzoquinone, naphthoquinones, phthalide derivative, furan naphthoquinones, fatty acid, sterols, phenolic compounds, flavonoids, carotenoids, glycoside, sugars, chalcone, ferulic esters and dihydroisocoumarins are reported in this family (Figure 2).
Terpenoids, including compounds 1–18 were isolated from Xantolis boniana var. boniana (twig and leaf extract), A. pulchra Sims (aerial part extract), A. triplinervia (leaf extract), N. laevis (root extract), C. grandiflora (flower extaract), Z. Montana (leaf extract), A. samydoides (leaf and stem extract), J. Decurrens (leaf extract), Macfadyena unguis-cati (liana extract), K. africana (leaf and fruit extract) and H. adenophyllum (leaf extract). Sterols and their derivatives: isolated from R. boniana (twig and leaf extract), R. xylocarpa (root extract), A. samydoides (leaf and stem extract), K. pinnatu (roots and bark extract) and Macfadyena unguis-cati (liana extract) are numbered 19-23 in Table 1.
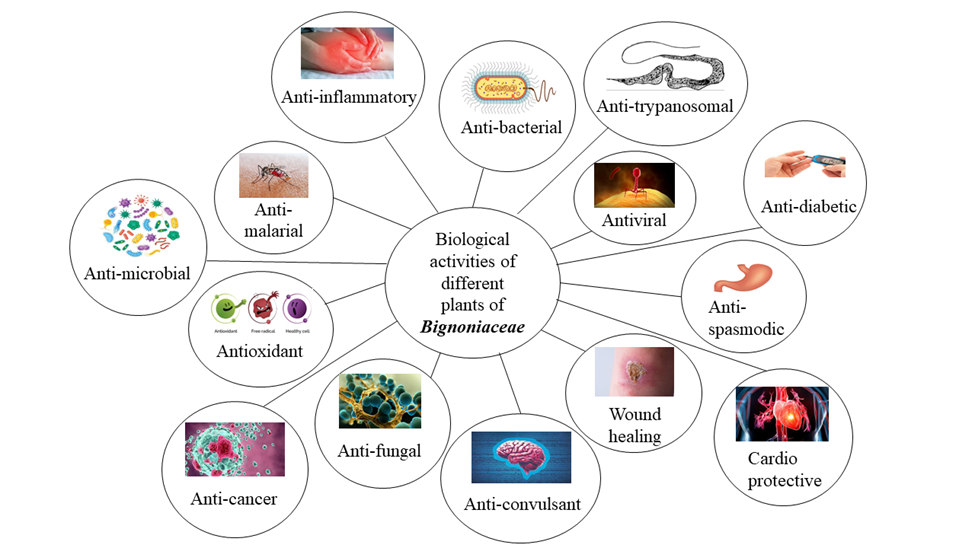
Figure 2: A Schematic Diagram of Biological Activities of Different Plants of Bignoniaceae
- Anthraquinones: isolated from S. zenkeri (stem-bark extract) and T. impetiginosa (bark extract) are numbered 24-27 in Table 1
- Benzoquinone: only one compound, numbered 28 was isolated from N. laevis (root extract)
- Naphthoquinones: isolated from K. pinnatu (root and fruit extract), N. laevis (root and stem-bark extract), T. undulate (stem-bark extract), T. avellanedue (stem-bark and whole plant extract), H. adenophyllum (stem and heartwood extract) and C. ovate (stem-bark extract) are numbered 29-53 in Table 1.
- Phthalide derivative: only one compound, numbered 54 was isolated from C.ovate (stem-bark extract).
- Furanonaphthoquinones: isolated from N. laevis (stem-bark extract), T. ochracea (stem-bark extract) and T.impetiginosa (bark extarct) are numbered 55-61 in Table 1.
- Phenolic compounds: isolated from A. patellifera (aerial part extract), Macfadyena unguis-cati (liana extract), N. laevis (stem-bark extarct), K. pinnatu (roots and fruit extract), S. zenkeri (stem-bark extract), K. africana (stem-bark extract), T. aurea (Stem-bark extract) and A. pulchra (aerial part extract) are numbered 62-69 in Table 1
- Fatty acids: isolated from Macfadyena unguis-cati (aerial part and seed extract) and Pyrostegia venusta (flower extract) are numbered 70-90 in Table 1
- Flavonoids: isolated from A. samydoides (leaves and stem extract), A. brachypoda (leaf and root extract), Z. tuberculosa (stem extract), A. pulchra (aerial part extarcat), M. hortensis (leaf and flower extract), O.indicum (stem-bark extract), N. laevis (root and stem-bark extract), Macfadyena unguis-cati (liana extract), A. chica (leaf extract) and T. stans (fruit extract) are numbered 91-108 in Table 1
- Carotenoids: isolated from A. chica (leaf extract) are numbered 109 and 110 in Table 1
- Glycoside: isolated from S. cylindricum (leaf and branch extract), M. stipulate (leaf and branch extract), F. adenophylla (leaf and branch extract), J. cuspidifolia (bark extarct), N. laevis (stem-bar extarct), Barnettia kerrii (Leaf and branch extract) and A. pulchra (leaf exatrct) are numbered 111-123 in Table 1.
- Dihydroisocoumarins: isolated from K. pinnatu (root and bark extract) are numbered 124-126 in Table 1
- Lignans: only one compound, numbered 127 was isolated from Kigelia Africana (leaf and fruit extract)
- Sugars: only one compound, numbered 128 was isolated from Tecoma stans (fruit extract)
- Alkaloids: isolated from Tecoma stans (leaf and fruit extract) are numbered 129-132 in Table 1
Table 1: Phytochemical Compounds of Family Bignoniaceae
|
Structure No |
Compound Name |
Source Type |
Part used |
Reference. |
Structures |
|
Terpenoids |
|||||
|
|||||
|
1 |
Bonianic acid A |
Radermachera boniana |
Twig and leaf extract |
[70] |
|
|
2 |
Bonianic acid B |
Radermachera boniana |
Twig and leaf extract |
[70] |
|
|
3 |
3-O-acetyluncaric acid |
Radermachera boniana |
Twig and leaf extract |
[70] |
|
|
4 |
Oleanolic acid |
Radermachera boniana, Arrabidaea pulchra, Arrabidaea triplinervia, Newbouldia laevis, Campsis grandiflora |
Twig and leaf extract Aerial part extract Leaf extract Root extract Flower extract |
[36,70,71,72] |
|
|
5 |
3-epioleanolic acid |
Radermachera boniana |
Twig and leaf extract |
[70] |
|
|
6 |
Arjunolic acid |
Campsis grandiflora |
Flower extract |
[72] |
|
|
7 |
Corosolic acid |
Campsis grandiflora |
Flower extract |
[72] |
|
|
8 |
Maslinic acid |
Campsis grandiflora |
Flower extract |
[72] |
|
|
9 |
Ursolic acid |
Radermachera boniana, Zeyheria Montana, Arrabidaea samydoides, Arrabidaea triplinervia, Jacaranda Decurrens, Campsis grandiflora |
Twig and leaf extract Leaf extract Leaf and stem extract Leaf extract Leaf extract Flower extract |
[70,71,73,74] |
|
|
10 |
Lupeol |
Arrabidaea samydoides, Macfadyena unguis-cati Kigelia Africana |
Leaf and stem extract Liana extract Leaf extract |
[71,75,76] |
|
|
11 |
Uvaol |
Arrabidaea samydoides |
Leaf and stem extract |
[71] |
|
|
12 |
Erythrodiol |
Arrabidaea samydoides |
Leaf and stem extract |
[71] |
|
|
13 |
Pomolic acid |
Arrabidaea triplinervia, Kigelia africana |
Leaf extract Leaf extract
|
[71,76] |
|
|
14 |
β-amyrin |
Haplophragma adenophyllum |
Leaf extract |
[77] |
|
|
15 |
Fibrarecisin |
Kigelia africana |
Leaf extract |
[76] |
|
|
16 |
Betulin |
Pyrostegia venusta |
Flower extract |
[78] |
|
|
17 |
Betulinic acid |
Pyrostegia venusta |
Flower extract |
[78] |
|
|
18 |
Canophyllol |
Kigelia africana |
Leaf and fruit extract |
[76] |
|
|
Sterols and their derivatives |
|||||
|
19 |
Ergosterol peroxide |
Radermachera boniana |
Twigs and leaf extract |
[70] |
|
|
20 |
Stigmasterol |
Radermachera xylocarpa, Arrabidaea samydoides Kigelia pinnatu |
Root extract Leaf and stem extract Roots and bark extract |
[71,79,80] |
|
|
21 |
Sitosterol |
Arrabidaea samydoides |
Leaf and stem extract |
[71] |
|
|
22 |
β-sitosterol |
Macfadyena unguis-cati Kigelia pinnatu |
Liana extract Roots and bark extract |
[75,80] |
|
|
23 |
β-sitostenone |
Radermachera boniana |
Twigs and leaf extract |
[70] |
|
|
Anthraquinones |
|||||
|
24 |
Zenkequinones A |
Stereospermum zenkeri |
Stem-bark extract |
[40] |
|
|
25 |
Zenkequinones B |
Stereospermum zenkeri |
Stem-bark extract |
[40] |
|
|
26 |
2-(hydroxymethyl) anthraquinone |
Tabebuia impetiginosa |
Bark extract |
[26] |
|
|
27 |
Anthraquinone-2-carboxylic acid |
Tabebuia impetiginosa
|
Bark extract |
[26] |
|
|
Benzoquinone |
|||||
|
28 |
2,3-dimethoxy-1,4-benzoquinone |
Newbouldia laevis |
Root extract |
[36] |
|
|
Naphthoquinones |
|||||
|
29 |
Kigelinone |
Kigelia pinnata |
Root and fruit extract |
[81] |
|
|
30 |
Isopinnatal |
Kigelia pinnata |
Root and fruit extract |
[81] |
|
|
31 |
Dehydro-α-lapachone |
Kigelia pinnata
|
Root and fruit extract |
[81] |
|
|
32 |
Lapachol |
Kigelia pinnata, Newbouldia laevis, Tecomella undulate, Tubebuiu avellanedue, Heterophragma adenophyllum |
Root and fruit extract Root extract Stem-bark extract Stem-bark extract Stem-heartwood extract |
[36,81,82,83,84] |
|
|
33 |
8-methoxydehydroiso-α-lapachone |
Catalpa ovate |
Stem-bark extract |
[85] |
|
|
34 |
9-methoxy-4-oxo-α-lapachone |
Catalpa ovate |
Stem-bark extract
|
[86] |
|
|
35 |
(4S,4aR,10R,10aR)-4,10-dihydroxy-2,2-dimethyl-2,3,4,4 α,10,10 α hexahydrobenzo[g]chromen-5-one |
Catalpa ovate |
Stem-bark extract
|
[86] |
|
|
36 |
3-hydroxydehydroiso-α-lapachone |
Catalpa ovate Heterophragma adenophyllum |
Stem-bark extract Heartwood extract |
[85,87] |
|
|
37 |
4,9-dihydroxy-α-lapachone |
Catalpa ovate |
Stem-bark extract |
[85]
|
|
|
38 |
4-hydroxy-α-lapachone |
Catalpa ovate |
Stem-bark extract |
[85] |
|
|
39 |
9-methoxy-α-lapachone |
Catalpa ovate |
Stem-bark extract |
[85] |
|
|
40 |
4-oxo-α-lapachone |
Catalpa ovate |
|
[86] |
|
|
41 |
5-hydroxy-dehydro-iso-α-lapachone |
Newbouldia laevis |
Stem-bark extract |
[88] |
|
|
42 |
3,8-dihydroxydehydroiso-α-lapachone |
Heterophragma adenophyllum |
Heartwood extract |
[87] |
|
|
43 |
α-lapachone |
Catalpa ovate |
Stem extract |
[86] |
|
|
44 |
Catalponone |
Catalpa ovate |
Stem extract |
[86] |
|
|
45 |
Catalponol |
Catalpa ovate |
Stem extract |
[86] |
|
|
46 |
β-Lapachone |
Tabebuia avellanedae |
Whole plant extract |
[86] |
|
|
47 |
Dilapachone |
Haplophragma adenophyllum |
Heartwood extract |
[77] |
|
|
48 |
Adenophyllone |
Haplophragma adenophyllum |
Heartwood extract |
[77] |
|
|
49 |
2-acetyl-4H,9H-naphtho[2,3-b]furan4,9-dione |
Tabebuia serratifolia
|
Bark extract
|
[26] |
|
|
50 |
2-(1-hydroxyethyl)-4H,9H-naphtho[2,3-b] furano-4,9-dione |
Tabebuia serratifolia |
Bark extract |
[26] |
|
|
51 |
4-Hydroxy-2-(3-hydroxy-2-methoxy-3-methyl-butylidene)- 3,4-dihydro-2H-naphthalen-1-one |
Catalpa ovate |
Stem extract |
[86] |
|
|
52 |
α-xiloidone |
Tabebuia avellanedae |
Wood extract |
[29] |
|
|
53 |
Epicatalponol |
Catalpa bignonioides |
Heartwood extract |
[89] |
|
|
Phthalide derivative |
|||||
|
54 |
Catalpalactone |
Catalpa ovate |
Stem-bark extract |
[85] |
|
|
Furanonaphthoquinones |
|||||
|
55 |
5-hydroxy-dehydroiso-α-lapachone |
Newbouldia laevis |
Stem-bark extract |
[88] |
|
|
56 |
2-isopropenylnaphtho[2,3-b]furan-4,9-dione |
Newbouldia laevis |
Stem-bark extract |
[88] |
|
|
57 |
2-(1'-methylethenyl)- 5-hydroxynaphtho[2,3-b]furan-4,9-dione |
Newbouldia laevis |
Stem-bark extract |
[88]
|
|
|
58 |
2-(1'-methylethenyl)-7-hydroxy-naphtho[2,3-b]furan-4,9-dione |
Newbouldia laevis |
Stem-bark extract |
[88] |
|
|
59 |
5,8-Dihydroxy-2-(1′-hydroxyethyl)naphtho[2,3-b]- furan-4,9-dione |
Tabebuia ochracea |
Stem-bark extract |
[90] |
|
|
60 |
8-Hydroxy-7-methoxy-2-acetylnaphtho[2,3-b]furan-4,9-dione |
Tabebuia ochracea |
stem bark extract |
[90] |
|
|
61 |
5-Hydroxy-2-( 1 -hydroxyethyl)naphtho[2,3-b] furan-4,9-dione |
Tabebuia impetiginosa |
Bark extract |
[91] |
|
|
Phenolic compounds |
|||||
|
62 |
p-Coumaric acid |
Kigelia pinnata, Arrabidaea pulchra, Stereospermum zenkeri |
Root and fruit extract Aerial part extract Stem-bark extract |
[40,71,81] |
|
|
63 |
Ferulic acid |
Kigelia pinnata |
Root and fruit extract |
[81] |
|
|
64 |
Chlorogenic acid |
Macfadyena unguis-cati |
Liana extract |
[75] |
|
|
65 |
Caffeic acid |
Kigelia pinnata |
Root and fruit extract |
[81] |
|
|
66 |
Atraric acid |
Newbouldia laevis |
Stem-bark extract |
[88] |
|
|
67 |
Atranorin |
Kigelia africana |
Stem-bark extract |
[92] |
|
|
68 |
Veratric acid |
Tabebuia aurea |
Stem-bark extract |
[38] |
|
|
69 |
Mangiferin |
Arrabidaea patellifera |
Aerial part extract |
[71] |
|
|
Fatty acids |
|||||
|
|||||
|
70 |
Dodecanoic acid (Lauric acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
71 |
Tetradecanoic acid (Myristic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
72 |
Pentadecenoic acid (Pentadecylic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
73 |
Arachidonic acid (Eicosanoic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
74 |
Heneicosylic acid (Heneicosanoic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
75 |
Behenic acid (Docosanoic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
76 |
Tricosylic acid (Tricosanoic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
77 |
Lignoceric acid (Tetracosanoic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
78 |
Pentacosylic acid (Pentacosanoic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
79 |
Cerotic acid (Hexacosanoic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
80 |
Montanic acid (Octacosanoic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
81 |
Melissic acid (Triacontanoic acid) |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
82 |
Pentadecanoic acid, 14- methyl |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
83 |
Heptadecanoic acid, 14 methyl |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
84 |
Hexadecanoic acid,14-methyl |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
85 |
Heptadecanoic acid,16-methyl |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
|||||
|
86 |
Palmitoleic acid |
Macfadyena unguis-cati |
Seed extract |
[93] |
|
|
87 |
Cis-vaccenic acid |
Macfadyena unguis-cati |
Seed extract |
[93] |
|
|
88 |
11-hexadecenoic acid |
Macfadyena unguis-cati |
Aerial part extract |
[93] |
|
|
89 |
Oleic acid |
Pyrostegia venusta |
Flower extract |
[78] |
|
|
|||||
|
90 |
Linoleic acid |
Pyrostegia venusta |
Flower extract |
[78] |
|
|
Flavonoids |
|||||
|
|||||
|
91 |
Chrysin |
Arrabidaea samydoides, Oroxylum indicum |
Leaf and stem extract Stem-bark extract |
[71,94] |
|
|
92 |
Cirsiliol |
Arrabidaea brachypoda |
Leaf extract |
[71] |
|
|
93 |
5,6,7,8-Tetramethoxyflavone |
Zeyheria tuberculosa |
Stem extract |
[41] |
|
|
94 |
5,6,7-Trimethoxyflavone |
Zeyheria tuberculosa |
Stem extract |
[41] |
|
|
95 |
4’-Hydroxy-5,6,7,8-tetramethoxyflavone |
Zeyheria tuberculosa |
Stem extract |
[41] |
|
|
96 |
4’-Hydroxy-5,6,7-trimethoxyflavone |
Zeyheria tuberculosa |
Stem extract |
[41] |
|
|
97 |
Hispidulin
|
Arrabidaea brachypoda, Millingtonia hortensis |
Leaf extract Leaf and flower extract |
[71,95]
|
|
|
98 |
Hortensin |
Millingtonia hortensis |
Flower extract |
[95] |
|
|
99 |
Apigenin |
Arrabidaea brachypoda, Newbouldia laevis |
Leaf extract Stem-bark extract |
[71,76] |
|
|
100 |
Luteolin |
Arrabidaea brachypoda, Newbouldia laevis |
Leaf and root extract Stem bark extract |
[71,76] |
|
|
101 |
Scutellarein |
Arrabidaea chica Oroxylum indicum |
Leaf extract Stem-bark extract |
[71,94] |
|
|
102 |
Chrysoeriol |
Newbouldia laevis |
Root extract |
[36] |
|
|
103 |
6-hydroxyluteolin-7-O-ß-glucoside |
Arrabidaea pulchra |
Aerial part extract |
[71] |
|
|
104 |
Baicalein |
Oroxylum indicum |
Stem-bark extract |
[94] |
|
|
105 |
6-Methoxyluteolin (Nepetin) |
Oroxylum indicum |
Stem-bark extract |
[94] |
|
|
|||||
|
106 |
Rutin |
Arrabidaea brachypoda Tecoma stans |
Leaf extract Fruit extract |
[71,97] |
|
|
107 |
Kaempferol |
Arrabidaea chica |
Leaf extract |
[71] |
|
|
108 |
Quercitrin |
Macfadyena unguis-cati |
Liana extract |
[75] |
|
|
Carotenoids |
|||||
|
109 |
α-carotene |
Arrabidaea chica |
Leaf extract |
[71] |
|
|
110 |
β-carotene |
Arrabidaea chica |
Leaf extract |
[71] |
|
|
Glycoside |
|||||
|
|||||
|
111 |
Stereospermoside |
Stereospermum cylindricum |
Leaf and branch extract |
[98] |
|
|
112 |
Ajugol |
Stereospermum cylindricum |
Leaf and branch extract |
[98] |
|
|
113 |
Specioside |
Stereospermum cylindricum |
Leaf and branch extract |
[98] |
|
|
114 |
Verminoside |
Stereospermum cylindricum |
Leaf and branch extract |
[98] |
|
|
|||||
|
115 |
Verbascoside |
Arrabidaea pulchra, Jacaranda cuspidifolia, Newbouldia laevis |
Leaf extract Bark extract Stem-bark extract |
[23,71,88] |
|
|
116 |
Isoverbascoside |
Stereospermum cylindricum |
Leaf and branch extract |
[98] |
|
|
117 |
Markhamiosides A |
Markhamia stipulate |
Leaf and branch extract |
[20] |
|
|
118 |
Salidroside |
Fernandoa adenophylla |
Leaf and branch extract |
[21] |
|
|
119 |
Leucosceptoside A |
Fernandoa adenophylla |
Leaf and branch extract |
[21] |
|
|
120 |
Martynoside |
Fernandoa adenophylla |
Leaf and branch extract |
[21] |
|
|
|||||
|
121 |
Khaephuoside A |
Barnettia kerrii |
Leaf and branch extract |
[99] |
|
|
122 |
khaephuoside B |
Markhamia stipulate |
Leaf and branch extract |
[20] |
|
|
123 |
Seguinoside K |
Barnettia kerrii |
Leaf and branch extract |
[99] |
|
|
Dihydroisocoumarins |
|||||
|
124 |
Kigelin |
Kigelia pinnatu |
Root extract |
[80] |
|
|
125 |
6-demethylkigelin |
Kigelia pinnatu |
Root extract |
[80] |
|
|
126 |
6-methoxymellein |
Kigelia pinnate |
Root and bark extract |
[80] |
|
|
Lignan |
|||||
|
127 |
Sesamin |
Kigelia Africana |
Leaf and fruit extract |
[76] |
|
|
Sugars |
|||||
|
128 |
Sucrose |
Tecoma stans |
Fruit extract |
[97] |
|
|
Alkaloids |
|||||
|
129 |
5b-Hydroxyskitanthine |
Tecoma stans |
Leaf extract |
[100] |
|
|
130 |
7-hydroxydehydroskytanthine |
Tecoma stans |
Fruit extract |
[100] |
|
|
131 |
4-hydroxytecomanine |
Tecoma stans |
Fruit extract |
[100] |
|
|
132 |
Tecomanine |
Tecoma stans |
Fruit extract |
[100] |
|
Biological Properties
The Bignoniaceae family has medicinal value because it contains secondary metabolites like fatty acids, glycosides, terpenoids, phenolic compounds naphthoquinones, alkaloids, flavonoids, and carotenoids. The biological activities of numerous species of the Bignoniaceae family are shown below in Table 2.
Table 2: Biological Activity of Species of Family Bignoniaceae
|
Biological activity |
Plant sources |
Inferences |
Reference. |
|
Antibacterial activity
|
Tabebuia ochracea |
At doses between 1.25 and 10 mg/well, an ethyl acetate extract of inner bark of T. ochracea prevents the formation of Staphylococcus aureus. |
[26,69] |
|
Kigelia pinnata |
The methanol extract of K. pinnata has the highest antibacterial activity against Proteus vulgaris and Salmonella typhi, intermediate antibacterial activity against S. aureus, E. coli, and B. cereus, but lower antibacterial activity against Klebsiella pneumonia, P. aeruginosa and Enterobacter aerogens. |
[27] |
|
|
Tabebuia rosea |
It has also been determined that the ethanol extract derived from leaves of T. rosea can stop Klebsiella pneumonia from growing at doses between 50,000 and 30,000 mg/L (50 and 300 mg/mL). |
[28] |
|
|
Jacaranda oxyphylla |
Ethanol extracts from J. oxyphyll leaves were discovered to have a relevant effect against Gram-positive bacteria (S. aureus and B. cereus). |
[37]
|
|
|
Tabebuia avellanedae |
T. avellanedae's hexane extract shown antibacterial effects against and methicillin-sensitive Staphylococcus aureus and methicillin-resistant Staphylococcus aureus. |
[29] |
|
|
Tecoma stans |
Leaf extract of Tecoma stans exhibit a potential broad spectrum antibacterial activity. |
[30] |
|
|
Pyrostegia venusta |
Pyrostegia venusta extract had antibacterial efficacy against Salmonella typhimurium , Staphylococcus aureus, Klebsiella pneumoniae, Pseudomonas aeruginosa, Bacillus cereus, Shigella sonnei and Escherichia coli. |
[22] |
|
|
Tabebuia impetiginosa |
Campylobacter jejuni was unaffected by T. impetiginosa hydro-alcoholic extract, which only had a 36% inhibitory effect on Helicobacter pylori growth. |
[32] |
|
|
Tabebuia chrysantha |
Dose of 125,000 mg/L (125 mg/mL) of methanolic extract of T. chrysantha leaf stopped the growth of Staphylococcus aureus. |
[33] |
|
|
Jacaranda mimosaefolia |
E. coli, S. typhi, B. cereus, and S. aureus are among the gram negative and gram positive bacteria that methanolic leaf extract from Jacaranda mimosaefolia is effective against. |
[34] |
|
|
Jacaranda acutifolia |
Xanthomonas campestris growth was suppressed by jacaranda acutifolia extract. |
[35] |
|
|
Jacaranda cuspidifolia |
J. cuspidifolia's methanolic extract shown antibacterial action against S. aureus, S. Pyogenes and N. gonorrheae. |
[23] |
|
|
Newbouldia laevis |
When compared to the standard antibiotics gentamycin and nystatin, Newbouldiaquinone A compound of Newbouldia laevis was 24 and 13 times more effective against Enterobacter aerogens and Candida gabrata, respectively. |
[36] |
|
|
Antimicrobial activity |
Tabebuia aurea |
The chemicals identified in T. aurea demonstrated a broad spectrum of efficacy against Gram negative and Gram positive bacteria and also against alcohol-acid bacteria and fungus in microbiological experiments |
[38] |
|
Kigelia pinnata |
Kigelia pinnata's crude aqueous extract shown strong antimicrobial activity. |
[39] |
|
|
Stereospermum zenkeri |
The best antibacterial activity against gram-negative Pseudomonas aeruginosa was demonstrated by the compound zenkequinone B of Stereospermum zenkeri (MIC 9.50 μg/mL). |
[40] |
|
|
Zeyheria tuberculosa |
Extracts and isolated flavones from Z. tuberculosa may be very effective against the pathogenic bacteria S. aureus and C. albicans. |
[41] |
|
|
Antifungal activity |
Tabebuia avellanedae |
Dichloromethane extract from bark of T. avellanedae has a significant antifungal effect, particularly against Microsporum gypseum, Aspergillus fumigatus, Saccharomyces cerevisiae, Candida albicans, and Trichophyton mentagrophytes. |
[42] |
|
Tabebuia caraiba |
Dose of 20,000 mg/L (20 mg/mL) of an ethanol extract from T. caraiba suppressed the growth of Candida albicans. |
[43] |
|
|
Tabebuia avellanedae |
Candida parapsilosis, Candida albicans, Candida krusei, Candida dubliniensis, Candida rugosa, Candida lusitaniae, and Candida glabrata are all inhibited by methanol extract from T. avellanedae, with MIC values ranging from 60 to 0.1 mg/L (0.06 to 0.0001 mg/mL). |
[44] |
|
|
Tecoma stans |
Extracts demonstrated antifungal efficacy against Candida albicans, Cryptococcus neoformans, and Microsporum gypsum. |
[45] |
|
|
Antiviral activity |
Tabebuia impetiginosa |
The extract of Tabebuia impetiginosa exhibited anti-HHV-1 action, with an EC50 of 166.6 µg/mL. |
[46] |
|
Antidiarrhoeal activity |
Kigelia pinnata |
When compared to atropine, the antidiarrheal impact of Kigelia pinnata at 500 mg/kg was found to be 82% and 62.7% on small intestine motility and castor oil-induced diarrhoea, respectively. |
[47] |
|
Anticancer activity |
Kigelia pinnata |
Methanolic extract of Kigelia pinnata significantly killed off human tumour cell lines. |
[48] |
|
Tabebuia avellanedae |
When taken orally, Tabebuia avellanedae's aqueous extract reduced the nociception caused by acetic acid. |
[50] |
|
|
Tecoma stans |
The leaf extract from Tecoma stans shown a considerable antiproliferative action. At concentrations of 7.8 g/mL and 1000 g/mL, extracts demonstrated minimum inhibition of 14.6% and maximal inhibition of 95.9%, respectively. |
[49] |
|
Kigelia pinnata |
Due to the existence of a high phenolic content, the ethyl acetate fraction of root of Kigelia pinnata exhibits excellent antioxidant activity against DPPH. It has strong antioxidant properties that prevent peroxidation of lipid, lower level of glutathione, and increase CAT and SOD activity. |
||
|
Pyrostegia venusta |
The fraction of ethyl acetate from Pyrostegia venusta roots demonstrates good antioxidant property against DPPH because it has a high phenolic content. |
[31] |
|
|
Tecoma stans |
Dose of 20 µg/mL of methanolic extract from T. stans demonstrated significant antioxidant activity. |
[51] |
|
|
Tabebuia impetiginosa |
The methanolic extract and syrup of Tabebuia impetiginosa had strongest antioxidant activity. |
[17] |
|
|
Antidiabetic activity |
Parmentiera edulis |
After being given to rats with alloxan-induced diabetes, the substance lactucin-8-O-methylacrylate of Parmentiera edulis reduces sugar levels in blood. |
[33] |
|
Tecoma stans |
Tecoma stans stimulates glucose absorption in both insulin-resistant and insulin-sensitive human and murine adipocytes, contributing to its anti-diabetic properties. |
[52] |
|
|
Kigelia pinnata |
The amylase inhibition assay was used to evaluate the anti-diabetic effect from leaf extract of Kigelia pinnata and results showed that it has strong efficacy against diabetes. |
[53] |
|
|
Anti-convulsant activity |
Spathodea campanulata |
Following oral administration, an ethanol extract of S. campanulata has anticonvulsant properties against PTZ, picrotoxin and MES-induced seizures in mice. |
[101] |
|
Kigelia pinnata |
PTZ (pentylene tetrazole) and MES (maximal 45 electro shock) induced convulsions were significantly prevented by methanolic extract of Kigelia pinnata. |
[54] |
|
|
Anti-plasmodial activity |
Spathodea campanulata |
Aqueous extract of leaf and the most polar portion of the chloroform extract of Spathodea campanulata showed a high amount of activity against Plasmodium herghei berghei in mice. |
[55] |
|
Anti-inflammatory activity |
Tabebuia avellanedae |
The expression of iNOS and COX-II, arachidonic acid-induced ear edoema and the production of NO and PGE2 were all inhibited by Tabebuia avellanedae water extract through preventing the phosphorylation of ERK. |
[59] |
|
Jacaranda decurrens |
Without inducing acute toxicity, jacaranda decurrens extract exhibits anti-inflammatory effects in rats. |
[58] |
|
|
Pyrostegia venusta |
30-300 mg/kg oral administration of hydroethanolic extract of Pyrostegia venusta showed an anti-inflammatory effect. Paw edoema was reduced and leukocyte migration into the peritoneal cavity was prevented by hydroethanolic extract of Pyrostegia venusta. |
[56] |
|
|
Kigelia pinnata |
The ethanolic extract of Kigelia pinnata's stem bark has been shown to have strong anti-inflammatory effects. |
[57] |
|
|
Tabebuia impetiginosa |
The generation of NO and PGE2, as well as the mRNA levels of COX-2 and iNOS was reduced by a water extract of Tabebuia impetiginosa. |
[17] |
|
|
Analgesic activity |
Stereospermum kunthianum |
The findings suggest that the analgesic activity from stem-bark of kunthianum in its aqueous extract is mediated by peripheral and central processes. |
[61] |
|
Kigelia pinnata |
K. pinnata leaf extract significantly reduced the pain caused by thermal noxious stimuli. |
[60] |
|
|
Larvicidal activity |
Millingtonia hortensis |
When tested on the three mosquito species Anopheles Stephensi, Aedes Aegypti, and Culex quinquefasciatus, acetone extract of the leaves of illingtonia hortensis was effective against all larval stages of these species. |
[62] |
|
Antinociceptive activity |
Pyrostegia venusta |
In Swiss male mice exposed to acetic acid-induced writhing, extracts of Pyrostegia venusta demonstrated antinociceptive action. |
[56] |
|
Cardioprotective effect |
Tecoma stans |
In a dose-dependent way, treatment with a 70% ethanolic extract of T. stans flowers has blocked the decline of GSH, SOD, and CAT levels. |
[63] |
|
Wound healing activity |
Pyrostegia venusta |
Pyrostegia venusta extract has a strong capacity for wound healing. Pyrostegia venusta extract was discovered to up-regulate TNF- α and IL-6 levels during the early stages of wound healing. |
[22] |
|
Tecoma stans |
In excision and incision wound models, Tecoma stans bark methanolic extract exhibits greater wound healing properties than chloroform and petroleum ether extracts. |
[64] |
|
|
Antispasmodic effect |
Tecoma stans |
Without involving β-adrenoceptors, opioid receptors, potassium channels, or NO generation, Tecoma stans leaf extract produces its antispasmodic actions. |
[65] |
|
Antimalarial activity |
Newbouldia laevis |
In vitro parasitic development of Plasmodium falciparum is moderately chemo-suppressed by the antimalarial compound Newbouldiaquinone A of Newbouldia laevis. |
[66] |
|
Kigelia pinnata |
The antimalarial efficacy of wood extract of Kigelia pinnata is superior to chloroquine and quinine against drug-resistant strains of Plasmodium falciparum. |
[67] |
|
|
Anti-trypanosomal activity |
Tabebuia pulcherrima |
After only 48 hours, leaf extract from T. pulcherrima revealed IC50 values of 7.8 g/mL. |
[68] |
|
Tabebuia pallida |
After only 48 hours, leaf extract from T. pallida revealed IC50 values of 7.2 g/mL. |
[68] |
|
|
Tabebuia rosea |
After 48 and 72 hours, an extract of the stem of T. rosea exhibited activity with IC50 of 13.4 and 16.2 g/mL, respectively. |
[68] |
|
|
Tabebuia serratifolia |
The chloroform extract of bark of T. serratifolia was reported to be effective against Trypa-nosoma cruzi with an inhibition percent greater than 96%. |
[69] |
Antibacterial Activity
At doses between 1.25 and 10 mg/well, an ethyl acetate extract of inner bark of T. ochracea prevents the formation of Staphylococcus aureus [26]. The methanol extract of K. pinnata has the highest antibacterial activity against Proteus vulgaris and Salmonella typhi, intermediate antibacterial activity against S. aureus, E. coli, and B. cereus, but lower antibacterial activity against Klebsiella pneumonia, P. aeruginosa and Enterobacter aerogens [27]. It has also been determined that the ethanol extract derived from leaves of T. rosea can stop Klebsiella pneumonia from growing at doses between 50,000 and 30,000 mg/L (50 and 300 mg/mL) [28]. T. avellanedae's hexane extract shown antibacterial effects against and methicillin-sensitive Staphylococcus aureus and methicillin-resistant Staphylococcus aureus [29]. Leaf extract of T. stans exhibit a potential broad spectrum antibacterial activity [30]. Pyrostegia venusta extract had antibacterial efficacy against Salmonella typhimurium , Staphylococcus aureus, Klebsiella pneumoniae, Pseudomonas aeruginosa, Bacillus cereus, Shigella sonnei and Escherichia coli [31]. Campylobacter jejuni was unaffected by T. impetiginosa hydro-alcoholic extract, which only had a 36% inhibitory effect on Helicobacter pylori growth [32]. Dose of 125,000 mg/L (125 mg/mL) of methanolic extract of T. chrysantha leaf stopped the growth of Staphylococcus aureus [33]. J. cuspidifolia's methanolic extract shown antibacterial action against S. aureus, S. Pyogenes and N. gonorrheae [23]. E. coli, S. typhi, B. cereus, and S. aureus are among the gram negative and gram positive bacteria that methanolic leaf extract from Jacaranda mimosaefolia is effective against [34]. Xanthomonas campestris growth was suppressed by j. acutifolia extract [35]. When compared to the standard antibiotics gentamycin and nystatin, Newbouldiaquinone A compound of Newbouldia laevis was 24 and 13 times more effective against Enterobacter aerogens and Candida gabrata, respectively [36]. Ethanol extracts from J. oxyphyll leaves were discovered to have a relevant effect against Gram-positive bacteria (S. aureus and B. cereus) [37].
Antimicrobial Activity
The chemicals identified in T. aurea demonstrated a broad spectrum of efficacy against Gram negative and Gram positive bacteria and also against alcohol-acid bacteria and fungus in microbiological experiments [38]. Kigelia pinnata's crude aqueous extract shown strong antimicrobial activity [39]. The best antibacterial activity against gram-negative Pseudomonas aeruginosa was demonstrated by the compound zenkequinone B of Stereospermum zenkeri (MIC 9.50 μg/mL) [40]. Extracts and isolated flavones from Z. tuberculosa may be very effective against the pathogenic bacteria S. aureus and C. albicans [41].
Antifungal Activity
Dichloromethane extract from bark of T. avellanedae has a significant antifungal effect, particularly against Microsporum gypseum, Aspergillus fumigatus, Saccharomyces cerevisiae, Candida albicans, and Trichophyton mentagrophytes [42]. Dose of 20,000 mg/L (20 mg/mL) of an ethanol extract from T. caraiba suppressed the growth of Candida albicans [43]. Candida parapsilosis, Candida albicans, Candida krusei, Candida dubliniensis, Candida rugosa, Candida lusitaniae, and Candida glabrata are all inhibited by methanol extract from T. avellanedae, with MIC values ranging from 60 to 0.1 mg/L (0.06 to 0.0001 mg/mL) [44]. Extracts of Tecoma stans demonstrated antifungal efficacy against Candida albicans, Cryptococcus neoformans, and Microsporum gypsum [45].
Antiviral Aactivity
The extract from T. impetiginosa shown anti-HHV-1 action, with an EC50 value of 166.6 µg/mL [46].
Antidiarrhoeal
When compared to atropine, the antidiarrheal impact of Kigelia pinnata at 500 mg/kg was found to be 82% and 62.7% on small intestine motility and castor oil-induced diarrhoea, respectively [47].
Anticancer
Methanolic extract of Kigelia pinnata significantly killed off human tumour cell lines [48]. The leaf extract from Tecoma stans shown a considerable antiproliferative action. At concentrations of 7.8 g/mL and 1000 g/mL, extracts demonstrated minimum inhibition of 14.6% and maximal inhibition of 95.9%, respectively [49]. When taken orally, Tabebuia avellanedae's aqueous extract reduced the nociception caused by acetic acid [50].
Antioxidant Activity
Due to the existence of a high phenolic content, the ethyl acetate fraction of root of Kigelia pinnata exhibits excellent antioxidant activity against DPPH. It has strong antioxidant properties that prevent peroxidation of lipid, lower level of glutathione, and increase CAT and SOD activity. The fraction of ethyl acetate from Pyrostegia venusta roots demonstrates good antioxidant property against DPPH because it has a high phenolic content [31]. The methanolic extract and syrup of Tabebuia impetiginosa had strongest antioxidant activity [17]. Dose of 20 µg/mL of methanolic extract from T. stans demonstrated significant antioxidant activity [51].
Anti-Diabetic Activity
Tecoma stans stimulates glucose absorption in both insulin-resistant and insulin-sensitive human and murine adipocytes, contributing to its anti-diabetic properties [52]. The amylase inhibition assay was used to evaluate the anti-diabetic effect from leaf extract of Kigelia pinnata and results showed that it has strong efficacy against diabetes [53]. After being given to rats with alloxan-induced diabetes, the substance lactucin-8-O-methylacrylate of Parmentiera edulis reduces sugar levels in blood [33].
Anti-Convulsant Activity
After oral administration, an ethanol extract from S. campanulata has anticonvulsant properties against pentylene tetrazole (PTZ), picrotoxin and maximal 45 electro shock (MES) induced seizures in mice. PTZ and MES induced convulsions were significantly prevented by methanolic extract of Kigelia pinnata [54].
Antiplasmodial Activity
Aqueous extract of leaf and the most polar portion of the chloroform extract of Spathodea campanulata showed a high amount of activity against Plasmodium herghei berghei in mice [55].
Anti-Inflammatory
30-300 mg/kg oral administration of hydroethanolic extract of Pyrostegia venusta showed an anti-inflammatory effect. Paw edoema was reduced and leukocyte migration into the peritoneal cavity was prevented by hydroethanolic extract of Pyrostegia venusta [56]. The ethanolic extract of Kigelia pinnata's stem bark has been shown to have strong anti-inflammatory effects [57]. The generation of NO and PGE2, as well as the mRNA level of iNOS and COX-2 was reduced by a water extract of Tabebuia impetiginosa [17]. Without inducing acute toxicity, Jacaranda decurrens extract exhibits anti-inflammatory effects in rats [58]. The expression of iNOS and COX-II, arachidonic acid-induced ear edoema and the production of NO and PGE2 were all inhibited by Tabebuia avellanedae water extract through preventing the phosphorylation of ERK [59].
Analgesic Activity
K. pinnata leaf extract significantly reduced the pain caused by thermal noxious stimuli [60]. The findings suggest that the analgesic activity from stem-bark of Stereospermum kunthianum in its aqueous extract is mediated by peripheral and central processes [61].
Larvicidal Activity
When tested on the three mosquito species Anopheles Stephensi, Aedes Aegypti, and Culex quinquefasciatus, acetone extract of the leaves of illingtonia hortensis was effective against all larval stages of these species [62].
Antinociceptive Activity
In Swiss male mice exposed to acetic acid-induced writhing, extracts of Pyrostegia venusta demonstrated antinociceptive action [56].
Cardioprotective Effect
In a dose-dependent way, treatment with a 70% ethanolic extract of T. stans flowers has blocked the decline of GSH, SOD, and CAT levels [63].
Wound Healing Activity
Pyrostegia venusta extract has a strong capacity for wound healing. Extract was discovered to up-regulate TNF- α and IL-6 levels during the early stages of wound healing [22]. In excision and incision wound models, Tecoma stans bark methanolic extract exhibits greater wound healing properties than chloroform and petroleum ether extracts [64].
Antispasmodic Effect
Without involving β-adrenoceptors, opioid receptors, potassium channels, or NO generation, Tecoma stans leaf extract produces its antispasmodic actions [65].
Antimalarial
In vitro parasitic development of Plasmodium falciparum is moderately chemo-suppressed by the antimalarial compound Newbouldiaquinone A of Newbouldia laevis [66]. The antimalarial efficacy of wood extract of Kigelia pinnata is superior to chloroquine and quinine against drug-resistant strains of Plasmodium falciparum [67].
Anti-Trypanosomal Activity
After only 48 hours, leaf extract from T. pulcherrima revealed IC50 values of 7.8 g/mL. After only 48 hours, leaf extract from T. pallida revealed IC50 of 7.2 g/mL. After 48 and 72 hours, an extract of the stem of T. rosea exhibited activity with IC50 of 13.4 and 16.2 g/mL, respectively [68]. Chloroform extract of bark of T. serratifolia was reported to be effective against Trypanosoma cruzi with an inhibition percent greater than 96% [69].
Future perspective
Based on available data on medicinal plant of the Bignoniaceae family, these plants are used in treatment of disease like hepatitis, neuralgia, diabetes, backaches, syphilis, ulcers, abscesses, white skin patches, rheumatism, syphilis, gonorrhoea, and ulcers. This family can therefore have regarded as a significant family in folk medicinal practices. It is because of the presence of phytochemical constituents that have many effective biological activities, and this could potentially aid researchers in the discovery of novel natural drugs. Therefore, plants of Bignoniaceae family can become significant sources of novel drugs and lead compound. A clinical trial must be conducted to better understand their safety and efficacy. Other species that have not yet been investigated should be the subject of more research.
CONCLUSION
This review focuses on phytochemical components of various species of family Bignoniaceae and also highlights the importance of these species. The plants of this family possess high amounts of secondary metabolites such as anthraquinones, sterols, terpenoids, benzoquinone, flavonoids, naphthoquinones, furanonaphthoquinones, carotenoids, glycoside, and sugars. Overall 132 compounds of this family are highlighted in this review. Due to the presence of these compounds, species belonging to the family Bignoniaceae are considered to be an important resource for treating various ailments like antibacterial, antiviral, antifungal, antidiarrheal, anticancer, antioxidant, anticonvulsant, antidiabetic, analgesic, antimalarial, anti-trypanosomal, and cardioprotective.
Conflict of Interest
All authors declare no competing interests.
Credit Authorship Statement
M.Sajid Hamid Akash, participated in literature analysis and manuscript editing. Malik Saadullah, contributed to the conception of the study. Maryam Farrukh designed the main structure of the manuscript. Aiman Atiq and Hamza Sohail done the manuscript editing. All authors reviewed and approved the final version of manuscript.
REFERENCES
- Afrin, S. et al. "Phytochemical screening and evaluation of analgesic and thrombolytic activity of the crude methanolic extract of Calamus rotang L. leaves (Arecaceae)." International Journal of Pharmaceutical Sciences and Research, 2020.
- Kumar, A. et al. "Phytochemistry and pharmacology of Pyrostegia venusta: a plant of family Bignoniaceae." International Journal of Phytomedicine, 2013.
- Silva, R. et al. "Emerging source of bioactive compounds from Arecaceae family: a systematic review." Research, Society and Development, 2021.
- VB, A.B. et al. "Ethno-pharmacological potentialities of some ornamental plants in the family Bignoniaceae." Journal of Medicinal Plants Studies, 2015.
- Pramanik, J. et al. "In-vitro anti-inflammatory and anti-arthritic activity of arthrito." Journal of Pharmacognosy and Phytochemistry, 2018.
- Aishwarya, M. et al. "Antiarthritic activity of Murraya exotica Linn against formaldehyde induced arthritis in Wistar rats." Research Journal of Pharmaceutical, Biological and Chemical Sciences, 2018.
- Chinnasamy, V. et al. "Antiarthritic activity of Achyranthes aspera on formaldehyde-induced arthritis in rats." Open Access Macedonian Journal of Medical Sciences, 2019.
- Joselin, J. et al. "Phytochemical evaluation of Bignoniaceae flowers." Journal of Chemical and Pharmaceutical Research, 2013.
- De Andrade Lima, C.S. et al. "Cytotoxic pyranonaphthoquinones from Melloa quadrivalvis (Bignoniaceae)." Natural Product Research, 2005.
- Blatt, C.T. et al. "Flavonoids of Bignoniaceae from the cerrado and their possible taxonomic significance." Plant Systematics and Evolution, 1998.
- Kusmardiyani, S. et al. "Pyrostegia genus: an update review of phytochemical compounds and pharmacological activities." Biointerface Research in Applied Chemistry, 2021.
- Wang, M.Z. et al. "Bioactive constituents from Radermachera boniana." ChemistrySelect, 2016.
- Ali, R.M. et al. "Antifungal activity of some Bignoniaceae found in Malaysia." International Journal of Phytotherapy Research, 1998.
- Rahmatullah, M. et al. "An ethnomedicinal, pharmacological and phytochemical review of some Bignoniaceae family plants and their folk medicinal uses in Bangladesh." Advances in Natural and Applied Sciences, 2010.
- Munoz-Mingarro, D. et al. "Biological activity of extracts from Catalpa bignonioides Walt. (Bignoniaceae)." Journal of Ethnopharmacology, 2003.
- Sharma, C. and Ojha, N. "Phytochemistry of some plant species of the family Bignoniaceae: a review." Biospectra, 2016.
- Zhang, J. et al. "Tabebuia impetiginosa: a comprehensive review on traditional uses, phytochemistry, and immunopharmacological properties." Molecules, 2020.
- Prathibha, R. et al. "In vitro evaluation of antioxidant activity and estimation of total phenolic and flavonoid content of different extracts of Tecoma stans (L.)." International Journal of Pharmacy and Biology, 2018.
- Dhungana, B.R. et al. "Kigelia pinnata: exploration of potential medicinal usage in human ailments." Journal of Pharmaceutical Research, 2016.
- Kanchanapoom, T. et al. "Phenolic glycosides from Markhamia stipulata." Phytochemistry, 2002.
- Kanchanapoom, T. et al. "Lignan and phenylpropanoid glycosides from Fernandoa adenophylla." Phytochemistry, 2001.
- Roy, P. et al. "In vivo antioxidative, antimicrobial and wound healing activity of flower extracts of Pyrostegia venusta." Journal of Ethnopharmacology, 2012.
- Arruda, A.L.A. et al. "Jacaranda cuspidifolia Mart. (Bignoniaceae) as an antibacterial agent." Journal of Medicinal Food, 2011.
- Pham, T.N. et al. "Anti-arthritic activity and phytochemical composition of ‘Cao Khai’ (Coptosapelta flavescens Korth.)." Heliyon, 2022.
- Afifi, N.I. et al. "Phytochemical composition and antioxidant activity of two species related to family Arecaceae." Pharmaceutical Sciences Asia, 2022.
- Jiménez-González, F.J. et al. "Anti-infectious activity in plants of the genus Tabebuia." Universitas Scientiarum, 2013.
- Jeyachandran, R. and Mahesh, A. "Antimicrobial evaluation of Kigelia africana (Lam)." Research Journal of Microbiology, 2007.
- Sathiya, M. and Muthuchelian, K. "Studies on phytochemical profile and antibacterial activity of ethanolic leaf extract of Tabebuia rosea." Ethnobotanical Leaflets, 2008.
- Machado, T. et al. "In vitro activity of Brazilian medicinal plants and naphthoquinones against methicillin-resistant Staphylococcus aureus." International Journal of Antimicrobial Agents, 2003.
- Senthilkumar, C. et al. "In vitro antibacterial activity of crude leaf extracts from Tecoma stans and other plants against human pathogenic bacteria." International Journal of PharmTech Research, 2010.
- Roy, P. et al. "Preliminary study of the antioxidant properties of flowers and roots of Pyrostegia venusta." BMC Complementary and Alternative Medicine, 2011.
- Cwikla, C. et al. "Investigations into the antibacterial activities of phytotherapeutics against Helicobacter pylori and Campylobacter jejuni." International Journal of Phytotherapy Research, 2010.
- Perez, R. et al. "Hypoglycemic effects of lactucin-8-O-methylacrylate of Parmentiera edulis fruit." Journal of Ethnopharmacology, 2000.
- Adelanwa, E. and Habibu, I. "Phytochemical screening and antimicrobial activities of the methanolic leaf extract of Jacaranda mimosifolia and Sansevieria liberica." Tropical Bioscience, 2015.
- Mora, D.A. et al. "Active ingredients with bactericide properties extracted from Jacaranda acutifolia, Ruta chapelensis and Aloe vera." Revista Chapingo Serie Protección Vegetal, 1996.
- Eyong, K.O. et al. "Newbouldiaquinone A: a potential antimicrobial and antimalarial agent from Newbouldia laevis." Phytochemistry, 2006.
- Pereira, V. et al. "Antioedematogenic activity, acetylcholinesterase inhibition and antimicrobial properties of Jacaranda oxyphylla." Natural Product Research, 2016.
- Barbosa-Filho, J.M. et al. "Botanical study, phytochemistry and antimicrobial activity of Tabebuia aurea." Phyton, 2004.
- Akunyili, D.N. et al. "Antimicrobial activities of the stembark of Kigelia pinnata." Journal of Ethnopharmacology, 1991.
- Lenta, B.N. et al. "Anthraquinones from the stem bark of Stereospermum zenkeri with antimicrobial activity." Phytochemistry, 2007.
- Bastos, M.L.A. et al. "Antimicrobial activity and toxicity of Zeyheria tuberculosa extracts and constituents." Annals of Clinical Microbiology and Antimicrobials, 2009.
- Guiraud, P. et al. "Comparison of antibacterial and antifungal activities of lapachol and β-lapachone." Planta Medica, 1994.
- e Silva, F.M. et al. "Evaluation of the antifungal potential of Brazilian Cerrado medicinal plants." Mycoses, 2009.
- Höfling, J. et al. "Antimicrobial potential of plant extracts against Candida species." Brazilian Journal of Biology, 2010.
- Gandhi, M.I. and Ramesh, S. "Antifungal and haemolytic activities of extracts of Tecoma stans." Journal of Ecobiotechnology, 2010.
- Brandão, G.C. et al. "Antiviral activities of plants from Minas Gerais: screening Bignoniaceae species." Revista Brasileira de Farmacognosia, 2010.
- Owolabi, O. and Omogbai, E. "Antidiarrhoeal properties of Kigelia africana extract." Pharmacologyonline, 2009.
- Momekova, D. et al. "Antineoplastic activity of Kigelia pinnata stem bark extract." Journal of Cancer Therapy Research, 2012.
- Thirumal, M. et al. "In vitro anticancer activity of Tecoma stans on breast cancer cell line." International Journal of Pharma and Bio Sciences, 2012.
- De Miranda, F.G.G. et al. "Antinociceptive and antiedematogenic properties of Tabebuia avellanedae extract." BMC Clinical Pharmacology, 2001.
- Robinson, J.P. et al. "Antioxidant and cytotoxic activity of Tecoma stans against lung cancer cell line." Brazilian Journal of Pharmaceutical Sciences, 2017.
- Alonso-Castro, A.J. et al. "Antidiabetic plants Tecoma stans and Teucrium cubense and glucose uptake effects." Journal of Ethnopharmacology, 2010.
- Dhriti, V. et al. "Free radical scavenging and antidiabetic activity of Kigelia pinnata." World Journal of Pharmacy and Pharmaceutical Sciences, 2014.
- Singh, A. et al. "Anticonvulsant activity of Kigelia pinnata bark extract." International Journal of Pharmacy and Pharmaceutical Sciences, 2010.
- Makinde, J. et al. "Schizontocidal activity of Spathodea campanulata leaf extract on Plasmodium berghei." Phytotherapy Research, 1987.
- Veloso, C.C. et al. "Anti-inflammatory and antinociceptive effects of Pyrostegia venusta extract." Revista Brasileira de Farmacognosia, 2012.
- Amali, M. et al. "Analgesic and anti-inflammatory activities of Kigelia africana extract." Nigerian Journal of Pharmaceutical Sciences, 2012.
- Santos, J.A. et al. "Anti-inflammatory effects and toxicity of Jacaranda decurrens roots extract." Journal of Ethnopharmacology, 2012.
- Byeon, S.E. et al. "Anti-inflammatory effects of Tabebuia avellanedae extract." Journal of Ethnopharmacology, 2008.
- Namita, P. et al. "Evaluation of analgesic potential of Kigelia pinnata leaf extract." International Research Journal of Pharmacy, 2011.
- Ching, F.P. et al. "Analgesic activity of aqueous extract of Stereospermum kunthianum stem bark." Acta Poloniae Pharmaceutica, 2009.
- Kaushik, R. and Saini, P. "Larvicidal activity of leaf extract of Millingtonia hortensis against mosquito vectors." Journal of Vector Borne Diseases, 2008.
- Ittagi, S. et al. "Cardioprotective effect of hydroalcoholic extract of Tecoma stans flowers." Asian Pacific Journal of Tropical Disease, 2014.
- Das, C. et al. "Evaluation of methanolic bark extract of Tecoma stans for wound healing." International Journal of Pharmaceutical Technology, 2010.
- Gharib, N.M.K. et al. "Antispasmodic effect of Tecoma stans leaf extract on rat ileum." DARU Journal of Pharmaceutical Sciences, 2007.
- Chukwujekwu, J.C. et al. "Antibacterial, anti-inflammatory and antimalarial activities of Nigerian medicinal plants." South African Journal of Botany, 2005.
- Carvalho, L. et al. "In vitro activity of naphthoquinones against Plasmodium falciparum." Brazilian Journal of Medical and Biological Research, 1988.
- El-Hawary, S. et al. "DNA fingerprinting of Tabebuia species and anti-trypanosomal activity." Pharmaceutical Sciences Asia, 2022.
- González-Coloma, A. et al. "Antileishmanial and cytotoxic screening of Peruvian plants." Parasitology Research, 2012.
- Truong, N.B. et al. "Antituberculosis triterpenoids from Radermachera boniana." Journal of Natural Products, 2011.
- do Nascimento, J.R. et al. "Review of phytochemistry and pharmacology of the genus Arrabidaea." Pharmaceuticals, 2022.
- Kim, D.-H. et al. "Triterpenoids from Campsis grandiflora as enzyme inhibitors." Archives of Pharmacal Research, 2005.
- Guenka, L. et al. "Anti-inflammatory and antinociceptive effects of Zeyheria montana extract." Memórias do Instituto Oswaldo Cruz, 2008.
- Varanda, E. et al. "Effect of ursolic acid from Jacaranda decurrens." Journal of Natural Products, 1992.
- Duarte, D. et al. "Chemical characterization and biological activity of Macfadyena unguis-cati." Journal of Pharmacy and Pharmacology, 2000.
- Sidjui, L.S. et al. "Triterpenes and lignans from Kigelia africana." Journal of Applied Pharmaceutical Science, 2015.
- Jassbi, A.R. et al. "Novel naphthoquinones from Heterophragma adenophyllum." Helvetica Chimica Acta, 2004.
- Mostafa, N. et al. "Pyrostegia venusta: a botanical, pharmacological and phytochemical review." Medicinal and Aromatic Plants, 2013.
- Shetgiri, N. et al. "Radermachera xylocarpa as a source of lapachol." Acta Poloniae Pharmaceutica, 2001.
- Govindachari, T. et al. "Isolation of dihydroisocoumarins from Kigelia pinnata." Phytochemistry, 1971.
- Binutu, O.A. et al. "Antibacterial and antifungal compounds from Kigelia pinnata." Planta Medica, 1996.
- Khatri, A. et al. "Hepatoprotective activity of Tephrosia purpurea and Tecomella undulata." Journal of Ethnopharmacology, 2009.
- Wagner, H. et al. "Structure determination of naphthoquinones from Tabebuia avellanedae." Helvetica Chimica Acta, 1989.
- Joshi, K.C. et al. "Quinones from Haplophragma adenophyllum." Planta Medica, 1979.
- Fujiwara, A. et al. "Antitumor-promoting naphthoquinones from Catalpa ovata." Journal of Natural Products, 1998.
- Park, B.M. et al. "Naphthoquinones from Catalpa ovata and nitric oxide inhibition." Archives of Pharmacal Research, 2010.
- Khandelwal, P. et al. "Revision of stereochemistry of lapachone derivatives." Phytochemistry, 2014.
- Gormann, R. et al. "Furanonaphthoquinones from Newbouldia laevis." Phytochemistry, 2003.
- McDaniel, C. "Antitermitic components of southern catalpa heartwood." Journal of Chemical Ecology, 1992.
- Díaz, F. and Medina, J.D. "Furanonaphthoquinones from Tabebuia ochracea." Journal of Natural Products, 1996.
- Fujimoto, Y. et al. "Structure and stereochemistry of cytotoxic naphthoquinones from Tabebuia impetiginosa." Journal of the Chemical Society Perkin Transactions 1, 1991.
- Zofou, D. et al. "Antiplasmodial activity of Kigelia africana extracts." Parasitology Research, 2011.
- Aboutable, E. et al. "Phytochemical and bioactivity investigations of Macfadyena unguis-cati." International Journal of Biology and Biotechnology, 2007.
- Mohanta, B.C. et al. "Flavonoids from Oroxylum indicum." Natural Product Sciences, 2007.
- Chulasiri, M. et al. "Mutagenicity of flavonoids from Millingtonia hortensis." Environmental and Molecular Mutagenesis, 1992.
- Gormann, R. et al. "Newbouldiosides from Newbouldia laevis." Phytochemistry, 2006.
- Marzouk, M.S. et al. "Antioxidant and antiproliferative constituents of Tecoma stans." Natural Product Communications, 2006.
- Kanchanapoom, T. et al. "Glycosides from Stereospermum cylindricum." Phytochemistry, 2006.
- Kanchanapoom, T. et al. "Phenolic glycosides from Barnettia kerrii." Phytochemistry, 2002.
- Costantino, L. et al. "Isolation and pharmacological activities of Tecoma stans alkaloids." Il Farmaco, 2003.
- Ilodigwe, E.E. et al. "Anticonvulsant activity of Spathodea campanulata extract." Journal of Medicinal Food, 2010.

