Journal of Advanced Biological Sciences | Year 2025 | Volume 1 | Issue 1 | Pages 10-14
Evaluation of Serum NGAL as an Inflammatory Biomarker in Patients Undergoing Hemodialysis
Mohammed Jumaa Mohammed 1*, Entedhar Rifaat Sarhat2 and Mousa Mahmood Marbut31Medical Biochemistry, Kirkuk Health Directorate, Iraq
2Medical biochemistry, College of Medicine, Tikrit University, Iraq
3Medical physiology, College of Medicine, Tikrit University, Iraq
View PDF Download XML DOI: 10.66590/jabs.2025.01.01.03
Abstract
Background: Neutrophil gelatinase-associated lipocalin (NGAL) (lipocalin-2) is an endogenous bacteriostatic protein that is released from renal tubular epithelial cells, neutrophils, and macrophages. Aim: to investigate the role of serum Neutrophil Gelatinase-Associated Lipocalin (NGAL) among hemodialysis patients with chronic kidney disease (CKD). Materials and Methods: This prospective hospital-based study conducted in Kirkuk city. The study included 30 CKD patients undergoing regular hemodialysis at Kirkuk General Hospital, along with 30 healthy individuals as controls. Blood samples were collected before and after hemodialysis, as well as from the control group, and analyzed for various parameters. The blood samples were collected for determination of serum Neutrophil Gelatinase-Associated Lipocalin blood urea and serum creatinine levels were measured using biochemical colorimetric methods. Results: The findings of the study revealed that hemodialysis (HD) patients had significantly higher levels of serum neutrophil gelatinase-associated lipocalin (NGAL) compared to the control group. The average NGAL level in HD patients was 500.2±146.1 ng/ml, whereas it was 131.65±37.1 ng/ml in the control group. This suggests that NGAL may serve as a potential biomarker for inflammation in HD patients. Additionally, after the dialysis session, the serum NGAL levels in HD patients decreased to 457.2±120.1 ng/ml. This reduction indicates that the dialysis procedure may have a beneficial effect on reducing inflammation in HD patients, as reflected by the decrease in NGAL levels. Furthermore, the study observed elevated levels of urea and creatinine in the blood of chronic kidney disease (CKD) patients before undergoing hemodialysis. However, after the completion of the dialysis procedure, there was a significant decrease in these levels. This indicates the effectiveness of hemodialysis in removing waste products such as urea and creatinine from the bloodstream. Conclusions: These findings highlight the potential role of NGAL as an inflammatory biomarker in HD patients and demonstrate the efficacy of hemodialysis in reducing both NGAL levels and waste products in CKD patients. Further research and investigation are warranted to fully understand the clinical implications and the relationship between NGAL, inflammation, and the effectiveness of hemodialysis in managing CKD patients.
Introduction
Chronic kidney disease (CKD) describes abnormal kidney function and/or structure. It is common, frequently unrecognized and often exists together with other conditions (such as cardiovascular disease and diabetes) [1]. The risk of developing CKD increases with age. As kidney dysfunction progresses, some coexisting conditions become more common and increase in severity [2]. People with CKD are five to ten times more likely to die prematurely than they are to progress to end stage kidney disease. Many people are asymptomatic or have nonspecific symptoms such as lethargy, itch, or loss of appetite. Diagnosis is commonly made after chance findings from screening tests (urinary dipstick or blood tests), or when symptoms become severe [3]. One of the most important complications associated with CKD are dyslipidemia and cardiovascular risk [4]. Neutrophil gelatinase-associated lipocalin (NGAL) (lipocalin-2) is an endogenous bacteriostatic protein that is released from renal tubular epithelial cells, neutrophils, and macrophages [5-6]. NGAL is important as a host defense because it behaves as an antibacterial agent by scavenging bacterial siderophores to prevent bacteria from establishing an infection [7-8]. However, the positive association between tumor necrosis factor-alpha and the NGAL level is known. In addition, it has been reported that there is a correlation between systemic inflammation and NGAL level, which has been reported to be a prognostic factor predicting the severity of acute renal failure [9]. Plasma NGAL levels are increased in both acute kidney injury and CKD. Elevated plasma NGAL is a strong predictor of acute kidney injury, the subsequent need for acute renal replacement therapy and in-hospital mortality. Anemia in CKD patients is a multifactorial complex problem influenced by the combination of insufficient erythropoietin production, absolute and function iron deficiency, as well as chronic inflammatory states [10]. The aim of this study is to evaluate the role of serum NGAL in inflammation among hemodialysis patients
MATERIALS AND METHODS
This prospective hospital-based study was done in Kirkuk city from the period from the beginning of December 2022 to the end of March 2023. The study included 30 patients with CKD who underwent regular hemodialysis were included in this study at Kirkuk General Hospital and their age were between 18 to 80 years. They were clinically diagnosed by nephrologist as ESRD patients (on hemodialysis), based on their history, clinical examination, renal function tests and other laboratory tests, undergoing hemodialysis. The study also included 30 adult persons looks healthy with no prior medical or family history of CKD as a control participated in this study.
Inclusion Criteria
The main inclusion criteria were individuals ≥18 years of age with end stage CKD (GFR <15 (mL/min/1.73 m2). All subjects underwent a comprehensive medical health examination and filled out questionnaires on health and lifestyle at the time of enrollment.
Exclusion Criteria
Exclusion criteria for enrollment in the study were patients with acute chest infection, heart failure, history of lung TB, lung cancer and neuromuscular disease:
- Approval of the council of College of Medicine/ Tikrit University was obtain for the proposal of the study
- Approval permission was presented to the director of Kirkuk Health Directorate / Kirkuk General Hospital
- An interview was carried out with these patients using questionnaire form designed by the investigator including age, sex ,duration of CKD, duration of hemodialysis sessions, frequency of hemodialysis sessions per week and time of diagnosis of CKD
Five ml of blood was collected by vein puncture 5 ml syringes from each patient (before hemodialysis (pre —HD) and 2-4 hours after hemodialysis (post — HD) and control enrolled in this study. Blood samples were placed into two sterile test tubes, in one of them 1.5 ml of blood was put in test tube containing anticoagulant EDTA and used for assessment of complete blood count (CBC) test using Swelab autoanalyzer. The tube then were centrifuged (3000 rpm) for 15 min. The clear serum was pipetted into clear dry Eppendorf’s tubes and stored at (-20°C) for determination of neutrophil gelatinase associated lipocalin (NGAL) by ELISA.
RESULTS
The study findings revealed a significant difference in serum neutrophil gelatinase-associated lipocalin levels between hemodialysis patients and the control group. Specifically, hemodialysis patients exhibited higher serum neutrophil gelatinase-associated lipocalin levels (500.2±146.1 ng/ml) compared to individuals in the control group (131.65±37.1 ng/ml). The statistical analysis indicated a significant result with a p-value of 0.001 (Table 1).
The study revealed that patients with chronic renal disease exhibited the highest mean levels of blood urea (149.7±25.72 mg/dl) and serum creatinine (7.22 mg/dl) compared to the control group. In contrast, the control group showed the lowest mean levels of blood urea (8.51±0.75 mg/dl) and serum creatinine (0.71±0.18 mg/dl). The differences in mean levels between the two groups were highly significant, as indicated by a p-value of less than 0.01 (Table 2).
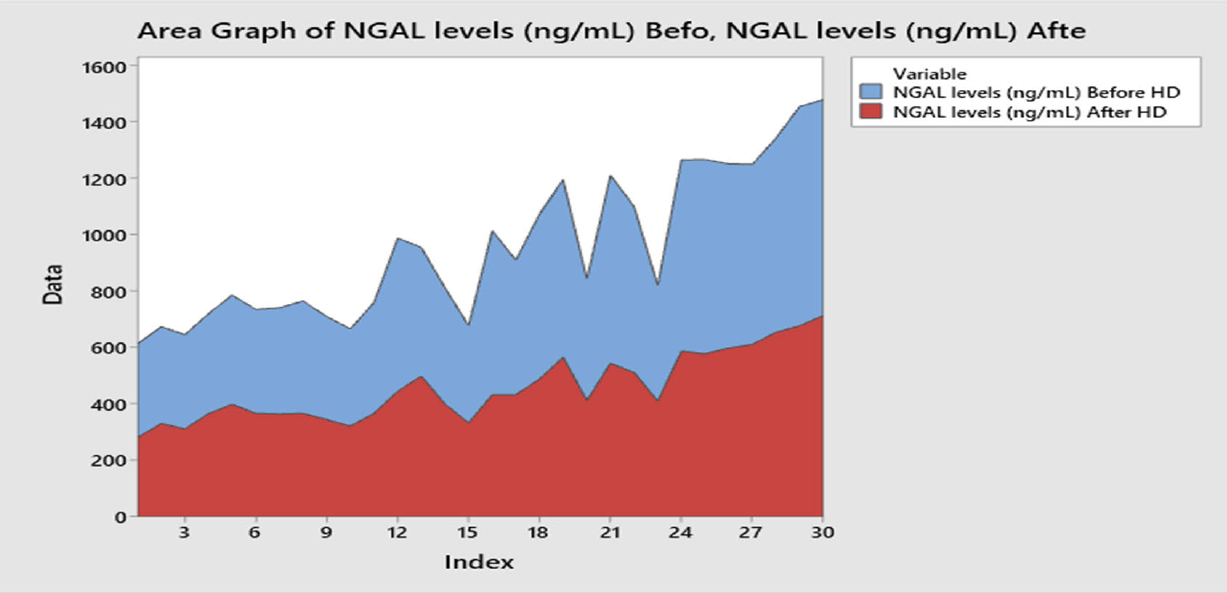
The study findings revealed that the mean serum NGAL (Neutrophil Gelatinase-Associated Lipocalin) levels before dialysis were 500.2±146.1 ng/ml, with a standard deviation of 500.2±146.1ng/ml. Following dialysis, the mean serum NGAL levels decreased to 457.2±120.1ng/ml, with a standard deviation of 120.1 ng/ml. The observed difference in NGAL levels before and after dialysis was found to be statistically significant (p:0.011), Table 3 and Figure 1.
The study showed that serum NGAL had positive correlation with serum ferritin, urea and creatinine and negative correlation with serum iron and hemoglobin level in patients before hemodialysis (r value> 0.2 and p-value<0.01) (Table 4).
Table 1: Mean of Serum Ngal in Hemodialysis Patients and the Control Group
|
Studied groups |
NGAL (ng/ml) |
p-value |
|||
|
Mean ±SD |
Min |
Median |
Max |
||
|
Hemodialysis patients |
500.2±146.1 |
331.9 |
443.5 |
777.1 |
0.001 |
|
Control group |
131.65±37.1 |
78.10 |
123.25 |
212.40 |
|
Table 2: Mean of Iron Markers in Hemodialysis Patients and the Control Group
|
Parameters (Mean±SD) |
Hemodialysis patients |
Control group |
p-value |
|
B. urea (mg/dl) |
149.7±25.72 |
29.5±7.8 |
0.001 |
|
S. creatinine (mg/dl) |
8.51±0.75 |
0.71±0.18 |
0.001 |
Table 3: Level of Serum Ngal in Patients Before and After Hemodialysis
|
Hemodialysis patients |
NGAL levels (ng/mL) |
p-value |
|||
|
Mean±SD |
Min |
Median |
Max |
||
|
Before dialysis |
500.2±146.1 |
331.9 |
443.5 |
777.1 |
0.011 |
|
After dialysis |
457.2±120.1 |
281.6 |
422.5 |
713.0 |
|
Table 4: Correlation of Hepcidin and Ngal with Other Parameters Before Hd
|
NGAL before dialysis |
Factors before dialysis |
r value* |
Type of correlation |
p-Value |
|
NGAL |
Ferritin |
0.55 |
Positive |
0.001 |
|
Iron |
-0.67 |
Negative |
0.001 |
|
|
B. Urea |
0.85 |
Positive |
0.001 |
|
|
Creatinine |
0.67 |
Positive |
0.001 |
|
|
TIBC |
0.04 |
No correlation |
0.71 |
|
|
Hb |
-0.84 |
Negative |
0.001 |
* r- value: Correlation coefficient, r- value: (+): positive correlation, (-): negative correlation, ≤0.2: No correlation (P-value >0.05), >0.2: correlation present (p-value ≤0.05)
DISCUSSION
In line with our finding, Aghsaeifard et al. [1] estimate the level of NGAL in 47 patients under chronic hemodialysis in end-stage renal disease (ESRD) and 15 healthy controls and recorded a significant elevation of NGAL concentration patients under chronic hemodialysis. Furthermore, studies have consistently demonstrated that neutrophil gelatinase-associated lipocalin (NGAL) levels are associated with the severity and progression of chronic kidney disease (CKD) and suggested that NGAL may serve as a valuable biomarker for assessing the extent of renal damage and underlying disease progression in CKD [2,3,4]. Moreover, Alderson et al. [5] found that blood NGAL levels in patients with chronic kidney disease (CKD) are an independent risk factor for progressing to end-stage renal disease (ESRD). They suggest that higher levels of NGAL in the blood are associated with an increased risk of ESRD and suggest that monitoring NGAL levels may aid in identifying patients at higher risk of developing ESRD, enabling timely intervention. In addition, Lobato et al. [6] conducted a study that compared the levels of NGAL in hemodialysis (HD) patients with those in control individuals. They found that HD patients had elevated levels of NGAL, indicating its potential as a biomarker in this population. The study also highlighted the predictive value of NGAL in assessing the progression of chronic kidney disease (CKD), particularly in the later stages. These findings suggest that NGAL can serve as a valuable tool for monitoring kidney function and disease progression in HD patients, aiding in the early detection and management of CKD [7]. Hemodialysis (HD) patients often experience ongoing renal injury and inflammation due to the underlying chronic kidney disease (CKD) and the process of dialysis itself. This sustained renal injury can lead to increased levels of neutrophil gelatinase-associated lipocalin (NGAL) in HD patients [8]. The NGAL is a protein that is produced and released by various cells, including renal tubular cells, in response to kidney injury and inflammation [9]. It is considered an early and sensitive biomarker for renal damage. In HD patients, the repeated exposure to dialysis, vascular access procedures, and potential complications such as infections can contribute to ongoing renal injury and inflammation [10]. The progression of kidney damage is characterized by elevated levels of two important chemical substances in the blood, namely creatinine and urea [11]. These substances are indicators of kidney function and are commonly used to assess Glomerular Filtration Rate (GFR) and renal function [12]. It's important to note that creatinine and urea are not directly toxic themselves but rather serve as measures of kidney function [13].
The findings of the current study are consistent with previous research. Al-Rawi et al. [14] reported a significant increase in the mean concentration of urea and creatinine in the studied groups compared to the control group. Similarly, Rasheed et al. [15] observed highly elevated serum creatinine levels in patients with chronic renal failure.
Chawla et al. demonstrated an increase in urea and creatinine levels in HD patients, which was associated with poorer functional renal recovery. Cheng et al. [15] also reported significantly elevated levels of blood urea and creatinine in HD patients.
Significantly elevated levels of blood urea and creatinine in hemodialysis (HD) patients can be attributed to several factors related to their underlying kidney disease and the process of dialysis itself. Firstly, HD patients have compromised kidney function, often due to end-stage renal disease (ESRD). Inadequate kidney function impairs the clearance of urea and creatinine, leading to their accumulation in the bloodstream [4,8]. Urea is a waste product produced by the breakdown of proteins, while creatinine is a byproduct of muscle metabolism. In healthy individuals with normal kidney function, these waste products are effectively filtered and eliminated through urine. However, in HD patients, the impaired kidney function results in reduced clearance and increased levels of urea and creatinine [16]. Secondly, the process of hemodialysis itself can contribute to elevated levels of blood urea and creatinine. During hemodialysis sessions, blood is circulated outside the body through a dialysis machine, which helps filter out waste products and excess fluid [17]. However, the efficiency of dialysis in completely removing urea and creatinine from the blood is not 100%. This can lead to residual levels of these waste products in the bloodstream even after a dialysis session, resulting in elevated levels [18]. The study findings indicate that there was a decrease in mean serum NGAL (Neutrophil Gelatinase-Associated Lipocalin) levels after dialysis compared to before dialysis. In line with these finding, Abd AlRahman et al. [18] study found that there was high statistically significant decrease in NGAL in dialyzed patients after dialysis when compared to the same patients before dialysis (p<0.001). Moreover, Reyes et al. [19] found that serum concentration of NGAL was significantly higher in patients that required versus did not require hemodialysis. Our results suggest that the dialysis procedure had an effect on NGAL levels in the study patients. The decrease in mean NGAL levels after dialysis implies a potential reduction in kidney injury or inflammation, as NGAL is known to be associated with these conditions [20,21]. The study found a positive correlation between serum NGAL and serum ferritin, urea, and creatinine levels in patients before hemodialysis. Additionally, there was a negative correlation between serum NGAL and serum iron and hemoglobin levels. These findings suggest that in patients before undergoing hemodialysis, elevated NGAL levels are associated with higher levels of ferritin, urea [22-24].
CONCLUSIONS
These findings highlight the potential role of NGAL as an inflammatory biomarker in HD patients and demonstrate the efficacy of hemodialysis in reducing both NGAL levels and waste products in CKD patients. Further research and investigation are warranted to fully understand the clinical implications and the relationship between NGAL, inflammation, and the effectiveness of hemodialysis in managing CKD patients.
REFERENCES
- Aghsaeifard Z, Alizadeh R and Bagheri N. “Association between neutrophil gelatinase-associated lipocalin (NGAL) and iron profile in chronic renal disease.” Archives of Physiology and Biochemistry, vol. 128, no. 3, 2022, pp. 703–707.
- Ding H et al. “Urinary neutrophil gelatinase-associated lipocalin (NGAL) is an early biomarker for renal tubulointerstitial injury in IgA nephropathy.” Clinical Immunology, vol. 123, 2017, pp. 227–234.
- Mertens C et al. “Macrophage-derived iron-bound lipocalin-2 correlates with renal recovery markers following sepsis-induced kidney damage.” International Journal of Molecular Sciences, vol. 21, no. 20, 2020, p. 7527.
- Ghosh S et al. “The role of lipocalin-2 in age-related macular degeneration (AMD).” Cellular and Molecular Life Sciences, 2020, pp. 835–851.
- Alderson HV et al. “The associations of blood kidney injury molecule-1 and neutrophil gelatinase–associated lipocalin with progression from CKD to ESRD.” Clinical Journal of the American Society of Nephrology, vol. 11, no. 12, 2016, pp. 2141–2149.
- Lobato GR, Lobato MR, Thomé FS and Veronese FV. “Performance of urinary kidney injury molecule-1, neutrophil gelatinase-associated lipocalin, and N-acetyl-β-D-glucosaminidase to predict chronic kidney disease progression and adverse outcomes.” Brazilian Journal of Medical and Biological Research, vol. 50, no. 5, 2017, p. e6106.
- Papathanasiou EA et al. “Perinatal lipocalin-2 profile at the extremes of fetal growth.” The Journal of Maternal-Fetal & Neonatal Medicine, vol. 34, no. 13, 2021, pp. 2166–2172.
- Urbschat A et al. “Macrophage-secreted lipocalin-2 promotes regeneration of injured primary murine renal tubular epithelial cells.” International Journal of Molecular Sciences, vol. 21, no. 6, 2020, p. 2038.
- Liu J, Sun B, Yin H and Liu S. “Hepcidin: a promising therapeutic target for iron disorders: a systematic review.” Medicine, vol. 95, no. 14, 2016, p. e3150.
- Daher R et al. “Heterozygous mutations in BMP6 pro-peptide lead to inappropriate hepcidin synthesis and moderate iron overload in humans.” Gastroenterology, vol. 150, no. 3, 2016, pp. 672–683.
- Komenda P, Yu N, Leung S et al. “Secular trends in end-stage renal disease requiring dialysis in Manitoba, Canada: a population-based study.” CMAJ Open, vol. 3, no. 1, 2015, pp. E8–E14.
- Sangeeta S et al. “Assessment of eGFR using cystatin-C and creatinine based equations for the early detection of renal injury in diabetic and non-diabetic patients.” Journal of Clinical & Diagnostic Research, vol. 12, no. 9, 2018, pp. 748–755.
- Oscanoa TJ et al. “Estimation of the glomerular filtration rate in older individuals with serum creatinine-based equations: a systematic comparison between CKD-EPI and BIS1.” Archives of Gerontology and Geriatrics, vol. 75, 2018, pp. 139–145.
- Al-Rawi KF et al. “Relationship between IL-2, IL-17 concentrations, and serum creatinine levels in men with chronic kidney diseases.” Reports of Biochemistry & Molecular Biology, vol. 10, no. 4, 2022, pp. 664–670.
- Rasheed N et al. “Serum hepcidin levels in anemia of chronic kidney diseases compared to iron deficiency anemia and its correlation with serum levels of HSC reactive protein, interleukin 6 and ferritin.” Global Journal of Biotechnology & Biochemistry, vol. 2, no. 1, 2013, pp. 43–50.
- Cheng Y et al. “Kidney impairment is associated with in-hospital death of COVID-19 patients.” MedRxiv, 2020, p. 2020-02.
- Liu YF et al. “The chronic kidney disease and acute kidney injury involvement in COVID-19 pandemic: a systematic review and meta-analysis.” PLoS One, vol. 16, no. 1, 2021, p. e0244779.
- Abd AlRahman A et al. “Serum neutrophil gelatinase-associated lipocalin as a marker of malnutrition in maintenance hemodialyzed patients.” Benha Medical Journal, vol. 38, no. 1, 2021, pp. 169–184.
- Reyes LF et al. “Serum levels of neutrophil gelatinase associated lipocalin (NGAL) predicts hemodialysis after coronary angiography in high risk patients with acute coronary syndrome.” BMC Nephrology, 2020, p. 506.
- Hassan HA. “Measurement of some biochemical values in hemodialysis patients in Baghdad.” Iraqi Journal of Pharmaceutical Sciences, vol. 23, no. 1, 2014, pp. 14–18.
- Omar AA, Aboud RR, Albakoush W and Anwesre RA. “Effect of ESRD on concentration of serum creatinine, urea and glucose in male patients.” MAYFEB Journal of Chemistry and Chemical Engineering, 2016, pp. 1–5.
- Bagalad BS et al. “Diagnostic accuracy of salivary creatinine, urea, and potassium levels to assess dialysis need in renal failure patients.” Dental Research Journal, vol. 14, no. 1, 2017, pp. 13–17.
- de Almeida ML et al. “Estimating the concentration of urea and creatinine in the human serum of normal and dialysis patients through Raman spectroscopy.” Lasers in Medical Science, vol. 31, no. 7, 2016, pp. 1415–1423.
- Singh S and Bhatta S. “Biochemical and hematological parameters in chronic kidney disease.” Journal of Manmohan Memorial Institute of Health Sciences, vol. 4, no. 1, 2018, pp. 4–11.Reference Two

