International Journal of Clinical and Medical Research | Year 2024 | Volume 1 | Issue 1 | Pages 1-30
Ethnopharmacology, Biological Properties, and Phytochemistry of Medicinally Important plant of Arecaceae Family: A Comprehensive review
Malik Saadullah1, Maryam Farrukh 2*1,3,4Department of Pharmaceutical Chemistry, Faculty of Pharmaceutical Sciences, Government College University, 38000, Faisalabad, Pakistan
2Department of Pharmacology, Faculty of Pharmaceutical Sciences, Government College University, 38000, Faisalabad, Pakistan
View PDF Download XML Download DOI XML DOI: 10.66590/ijcmr2024010101
Abstract
Ethnopharmacological relevance: The family Arecaceae is one of the largest plant families in the world. It is widely distributed in tropical regions (Brazil, Saudi Arabia, Middle Eastern countries and Egypt, Indonesia, Malaysia, and Thailand). Traditionally, plants of this family were used to treat various disorders, like gastrointestinal ailments, diabetes, spasms, sexual dysfunction, nephritis, rheumatism, cough, diarrhea, and hypertension. Aim of the review: This review highlights the biological properties, phytochemistry, and chemotaxonomic classification of the phytochemicals of different species of the family Arecaceae. Materials and methods: The relevant information on the family Arecaceae was collected from scientific databases (Google Scholar, ScienceDirect, ACS Publications, PubMed, Wiley Online Library). Information was also collected from online databases and books. theses. The literature cited in this review dates from 2001 to June 2022. Result: About 141 compounds have been isolated from different species of the family Arecaceae, including flavonoids, fatty acids, terpenoids, fatty acid esters, sugars, fatty alcohols, vitamins, carotenoids, tannins, ceramide derivatives, glyceryl derivatives, stilbenoid derivatives, simple phenolic glycosides, sterols, alkaloids, lignans, amino acids, and phenolic compounds. Their structure and presence in each species of this family are presented in tabular form. In biological studies, the crude extracts and metabolites of the medicinally important species of the family Arecaceae have different biological activities, including, antioxidant, antimicrobial, analgesic, renal protective, antiparasitic, cardioprotective, antidiabetic, anti-mutagenic, antiinflammatory, anticancer, diuretic, hepatoprotective, antihyperlipidemic, antipalatelet, antiviral, antipyretic, antifungal, antidiarrheal, antitrichomonal, antiacetylcholinestrase, and antihypertensive. This review examines folkloric uses, phytochemistry, and biological activities of selected members of this family.
INTRODUCTION
Herbal remedies, which have been employed in both traditional and modern systems since ancient times, are the earliest medical supplies that are known to exist. The WHO stated that herbal medicines are defined as herbs, herbal preparations, herbal finished products and herbal ingredients [1]. Around 80% of the world's population relies on herbal medicine for their primary healthcare requirements [2]. Patients favor herbal remedies over traditional ones due to a persistent illness and dread of surgery, severe morbidity, rising medical expenses and the drawbacks of innovative drugs [3].
Using plant components in their purest form, either fresh or dried, is a common practice in traditional medicine for treating and preventing a wide range of illnesses [4]. Alkaloids, flavonoids, phenolics, terpenoids and glycosides are some of the major types of therapeutic phytochemicals synthesized by plants [5]. Medicinal plants are used to find and develop new therapeutic medications [6].
Figure 1: Graphical Abstract

Figure 2: The Plants of Arecaceae Family, (a) Areca catechu, (b) Cocos nucifera, (c) Caryota urens, (d) Mauritia flexuosa, (e) Phoenix dactylifera and (f) Euterpe oleracea
The Arecaceae, also known as the Palm family (palmeae), is one of the most well-known plant families [7] (Figure 2). This family belongs to the monocot order Arecales of flowering plants [8]. It has 181 genera and 2600 species and it is one of the largest families in the world. Bush species, woody trees or even grasses are examples of the physical and functional diversity among this family's species. It is primarily found in tropical regions [9]. Species of this family are mostly present in Indonesia, Malaysia and Thailand [9-11].
According to previous phytochemical studies, this family contains anthocyanidins, lignans, benzenoids, benzoquinone, monoterpenoids and nor isoprenoids [12], fatty derivatives, sterols, phenolic acids and their derivatives, flavonoids and other classes [8].
Plants in this family have biological activities like hepatoprotective, anti-hyperlipidemic, anti-diabetic, anti-oxidant, anti-parasitic, antihypertensive, renal protective, cardioprotective, anti-microbial (antibacterial, antifungal and antiviral), anti-pyretic, anti-inflammatory, anti-mutagenic, anti-platelet, analgesic, anti-ulcer, neuropharmacological, anti-acetylcholinesterase, anti-Alzheimer [8] and anti-cancer activities [13].
Traditional Uses
The plants in this family are mostly used for medicinal purposes. According to a review of traditional medical practises, Chamaerops humilis is used as stipe or leaf extract for the treatment of gastrointestinal ailments, diabetes, spasms and digestive issues [14]. Regular use of Phoenix species has been traditionally used to treat sexual dysfunction, nephritis, rheumatism, burning feelings and cough [15]. Diarrhea is treated with a C. nucifera husk fibre extract in Brazil [16].
Herbal tea made from Hyphae thebaica is historically thought to be effective in treating hypertension [17]. Hypertension is treated with a decoction of the leaves of the Acrocomia aculeata plant [18].
The Colocasia gigantea tuber is used to alleviate drowsiness as well as to lessen "internal heat" (fever) [13].
Phyto-Constituents
Phytochemicals are biologically active, naturally occurring chemical substances that are present in plants and which enhance human health. Phytochemicals are plant compounds that shield plant cells from environmental dangers such dehydration, stress, UV exposure, pathogenic attack and pollution. Phytochemicals build up in a variety of plant tissues, including the roots, stems, leaves, flowers, fruits and seeds [19]. Tannins, anthraquinones, alkaloids, terpenoids, cardiac glycosides, steroids, flavonoids, phlobatannins, saponins and reducing sugars are the major phytochemical components found in therapeutic plants [20].
Compounds 1-142 were isolated from medicinally important genra of Arecaceae (Table 1). Commonly, phenolic compounds are the major constituents of this family. Many fatty acids, fatty alcohols, fatty acid esters, sterols, vitamins, carotenoids, tocopherols, phenolic compounds, amino acid, terpenoids, tannins, sugars, stilbenoid derivatives, glyceryl derivatives, alkaloids, lignan derivatives, ceramide derivatives and simple phenolic glycosides are reported in this family.
Flavonoids, including compounds 1-24, were isolated from H. indica (leaf extract), M. flexuosa (pulp and leaf extract), H. thebaica (fruit and epicarp extract), A. alexandrae (leaf extract), D. album (leaf extract), Caryota mitis (leaf extract), S. repens (pericarp extract), L. australis (leaf extract), S. edulis (peel extract), A. catechu (whole plant extract), R. rivularis (leaf extract), D. lutescens (whole plant extract ), H. verschaffeltii (leaf extract), A. alexandrae (leaf extract), C. urens (Base leaf extract), C. nucifera (fiber extract), P. dactylifera (fruit and pollen grain extract ), B. armata (fruit extract), S. zalacca (pulp extract) and W. robusta (leaf extract).
- Fatty Acids: Isolated from S. wallichiana (seeds), A. catechu (whole plant extract), M. flexuosa (pulp extract), P. dactylifera (whole plant), P. loureiroi (leaf extract), E. oleracea (fruit extract), L. australis (fruit extract) and A. aculeate (fruit extract) are numbered 25-35 in Table 1
- Fatty Acid Esters: Only one compound, numbered 36 was isolated from P. loureiroi (leaf extract)
- Fatty Alcohols: Only one compound, numbered 37 was isolated from P. loureiroi (leaf extract)
- Sterols: Isolated from S. wallichiana (root and fruit extract), A. catechu (whole plant extract), M. flexuosa (pulp extrcat), P. loureiroi (leaf extract), P. dactylifera (whole plant extract) and P. paludosa (leaf extract) are numbered 38-53 in Table 1
- Vitamins: Isolated from C. nucifera (liquid albumen extract), L. australis (pulp extract), M. flexuosa (pulp extract) and P. dactylifera (whole plant extract) are numbered 54-60 in Table 1
- Carotenoids: Isolated from M. flexuosa (fruit extrcat), numbered 61-63 in Table 1
- Tocopherols: Isolated from M. flexuosa (pulp extract), numbered 64-67 in Table 1
- Phenolic Compounds: Isolated from M. flexuosa (pulp extract), S. repens (pericarp extract), S. edulis (peel and pulp extract), R. rivularis (leaf extract), S. zalacca (pulp extract), L. chinensis (fruit extract), H. thebaica (fruit extract), C. nucifera (endocarp extract), A. catechu (whole plant extract ), W. robusta (leaf extract), P. paludosa (leaf extract) and B. armata (fruit extract) are numbered 68-98 in Table 1
- Amino Acids: Only one compound, numbered 99 was isolated from C. nucifera (Liquid albumen/solid albumen extract)
Terpenoids, including compounds 100-104, were isolated from R. rivularis (leaf extract), P. dactylifera (leaf extract) and P. paludosa (leaf extract).
- Tannins: Isolated from A. catechu (whole plant extract), numbered 105 and 106 in Table 1
- Stilbenoid Derivatives: Isolated from C. nucifera (Endocarp extract), P. dactylifera (stem extract) and A. aculeate (seed extract), numbered 107-113 in Table 1
- Glyceryl Derivatives: Only one monoacylglycerols compound, numbered 114 was isolated from L. chinensis (root extrcat)
- Alkaloids: Isolated from A. catechu (whole plant extract), numbered 115-124 in Table 1
- Lignan Derivatives: Isolated from C. quiquesetinervius (stem extract), numbered 125-132 in Table 1
- Ceramide Derivatives: Isolated from L. chinensis (root extract), numbered 133 and 134 in Table 1
- Sugars: Isolated from P. dactylifera (fruit extract) and P. paludosa (leaf extract), numbered 135-139 in Table 1
- Simple Phenolic Glycosides: Isolated from S. repens (pericarp extract), numbered 140 and 141 in Table 1
Table 1: Phytochemical Compounds of Family Arecaceae
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
Flavonoids |
|||||
|
1: Flavonol |
|||||
|
1 |
Kaempferol |
Hyophorbe indica, Mauritia flexuosa, Hyphaene thebaica |
Leaf extract Pulp extract Epicarp extract |
da Silva et al. [21] Pereira Freire et al. [22] Salib et al. [23] |
|
|
2 |
Rutin |
Archontophoenix alexandrae, Dictyosperma album, Mauritia flexuosa, Caryota mitis, Serenoa repens |
Leaf extract Leaf extract Leaf extract Leaf extract Pericarp extract |
Afifi et al. [12] Afifi et al. [12] Nonato et al. [24] El‐Akad et al. [25] Olennikov et al. [26] |
|
|
3 |
Avicularin |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
4 |
Myricetin |
Mauritia flexuosa |
Pulp extract |
Pereira Freire et al. [22] |
|
|
5 |
Quercetin |
Livistona australis, Salacca edulis |
Leaf extract Peel extract |
Kassem et al. [27] Kanlayavattanakul et al. [28] |
|
|
6 |
Rhamnetin |
Hyphaene thebaica |
Fruit extract |
Hussein et al. [29] |
|
|
7 |
Isorhamnetin |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
2: Flavone |
|||||
|
8 |
Apigenin |
Ravenea rivularis, Mauritia flexuosa, Hyphaene thebaica, Livistona australis |
Leaf extract Pulp extract Fruit extract Leaf extract |
da Silva et al. [21] Nonato et al. [24] Hussein et al. [29] Kassem et al. [27] |
|
|
9 |
Vitexin |
Hyphaene thebaica, |
Epicarp extract |
Salib et al. [23] |
|
|
10 |
Isovitexin |
Livistona australis, Hyphaene thebaica, Dypsis lutescens |
Leaf extract Epicarp extract Whole plant extract |
Kassem et al. [27] Salib et al. [23] Almaatty et al. [13] |
R1=R2=R4=R6=R7=H, R5=OH, R3=Glu |
|
11 |
Chrysoeriol |
Areca catechu, Hyphaene thebaica, |
Whole plant extract Epicarp extract |
Salehi et al. [30] Salib et al. [23] |
|
|
12 |
Acacetin |
Caryota urens |
Base leaf extract |
Mohammed and Fouad [31] |
|
|
13 |
Luteolin |
Hyophorbe verschaffeltii, Archontophoenix alexandrae, Ravenea rivularis, Mauritia flexuosa, Areca catechu, Hyphaene thebaica, Livistona australis |
Leaf extract Leaf extract Leaf extract Pulp extract Whole plant extract Epicarp extract Leaf extract |
Elgindi et al. [32] Afifi et al. [12] da Silva et al. [21] Pereira Freire et al. [22] Salehi et al. [30] Salib et al. [23] Kassem et al. [27] |
|
|
14 |
Tricin |
Archontophoenix alexandrae, Dictyosperma album, Livistona australis |
Leaf extract Leaf extract Leaf extract |
Afifi et al. [12] Afifi et al. [12] Kassem et al. [27] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
15 |
Isoorientin |
Mauritia flexuosa, Livistona australis |
Leaf extract Leaf extract |
Pereira Freire et al. [22] Kassem et al. [27] |
|
|
16 |
Orientin |
Mauritia flexuosa, Dictyosperma album, Livistona australis |
Leaf extract Leaf extract Leaf extract |
Pereira Freire et al. [22] Afifi et al. [12] Kassem et al. [27] |
|
|
17 |
Neodiosmin |
Phoenix dactylifera |
Fruit extract |
Fathy et al. [33] |
|
|
3: Flavanol |
|||||
|
18 |
Catechin |
Mauritia flexuosa, Cocos nucifera, Hyophorbe indica, Brahea armata, Hyphaene thebaica |
Pulp extract Fiber extract Leaf extract Fruit extract Fruit extract |
Nonato et al. [34] Lima et al. [16] da Silva et al. [21] Hussein et al. [36] Hussein et al. [29] |
|
|
19 |
Epicatechin |
Mauritia flexuosa, Hyophorbe indica, Salacca zalacca, Brahea armata |
Pulp extract leaf extract pulp extract Fruit extract |
Pereira Freire et al. [22] da Silva et al. [21] Saleh et al. [10] Hussein et al. [29] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
4: Flavanone |
|||||
|
20 |
Liquiritigenin |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
21 |
Eriocitrin |
Hyphaene thebaica |
Fruit extract |
Hussein et al. [29] |
|
|
22 |
Naringin |
Phoenix dactylifera |
Pollen grain extract |
Abbas and Ateya [37] |
|
|
23 |
Hesperetin |
Hyphaene thebaica |
Fruit extract |
Hussein et al. [29] |
|
|
24 |
Naringenin |
Washingtonia robusta |
Leaf extract |
Selim et al. [38] |
|
|
Fatty acids |
|||||
|
1: Saturated |
|||||
|
25 |
Lauric acid |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
26 |
Myristic acid |
Areca catechu, Mauritia flexuosa |
Whole plant extract Pulp extract |
Salehi et al. [30] Pereira Freire et al. [22] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
27 |
Stearic acid (Octadecanoic acid) |
Areca catechu, Mauritia flexuosa, Phoenix loureiroi |
Whole plant extract Pulp extract Leaf extract |
Salehi et al. [30] Pereira Freire et al. [22] Mondal et al. [15] |
|
|
28 |
Palmitic acid (n- Hexadecanoic acid) |
Euterpe oleracea, Acrocomia aculeate, Livistona australis, Mauritia flexuosa, Phoenix loureiroi |
Fruit extract Fruit extract Fruit extract Pulp extract Leaf extract |
da Silva et al. [21] da Silva et al. [21] Kassem et al. [39] Pereira Freire et al. [22] Mondal et al. [15] |
|
|
29 |
Arachidonic acid |
Mauritia flexuosa |
Pulp extract |
Pereira Freire et al. [22] |
|
|
2: Monounsaturated |
|||||
|
30 |
Oleic acid |
Euterpe oleracea, Acrocomia aculeate, Livistona australis, Phoenix dactylifera, Areca catechu, Mauritia flexuosa |
Fruit extract Fruit extract Fruit extract Whole plant extract Whole plant extract Pulp extract |
da Silva et al. [21] da Silva et al. [21] Kassem et al. [39] Ahmed et al. [11] Salehi et al. [30] Pereira Freire et al. [22] |
|
|
31 |
Palmitoleic acid |
Phoenix dactylifera, Euterpe oleracea, Mauritia flexuosa |
Whole plant extract Fruit extract Pulp extract |
Ahmed et al. [11] da Silva et al. [21] Pereira Freire et al. [22] |
|
|
32 |
Elaidic acid |
Mauritia flexuosa |
Pulp extract |
Pereira Freire et al. [22] |
|
|
3: Polyunsaturated |
|||||
|
33 |
Linoleic acid |
Euterpe oleracea, Acrocomia aculeate, Livistona australis, Salacca wallichiana, Phoenix dactylifera, Mauritia flexuosa |
Fruit extract Fruit extract Fruit extract Seeds extract Whole plant extract Pulp extract |
da Silva et al. [21] da Silva et al. [21] Kassem et al. [39] Ragasa et al. [40] Ahmed et al. [11] Pereira Freire et al. [22] |
|
|
34 |
Linolenic acid |
Euterpe oleracea, Phoenix dactylifera, Mauritia flexuosa |
Fruit extract Whole plant extract Pulp extract |
da Silva et al. [21] Ahmed et al. [11] Pereira Freire et al. [22] |
|
|
35 |
Margaric acid |
Mauritia flexuosa |
Pulp extract |
Pereira Freire et al. [22] |
|
|
4: Fatty Acid Esters |
|||||
|
36 |
Hexadecanoic acid ethyl ester |
Phoenix loureiroi, |
Leaf extract |
Mondal et al. [15] |
|
|
5: Fatty Alcohols |
|||||
|
37 |
2- Methyl Z, Z- 3,13- octadecadienol |
Phoenix loureiroi, |
Leaf extract |
Mondal et al. [15] |
|
|
Sterols |
|||||
|
38 |
Cholesterol |
Phoenix dactylifera |
Whole plant extract |
Ahmed et al. [11] |
|
|
39 |
Stigmasterol |
Phoenix dactylifera, Mauritia flexuosa, Phoenix loureiroi, Salacca wallichiana |
Whole plant extract Pulp extract Leaf extract Root extract |
Ahmed et al. [11] Pereira Freire et al. [22] Mondal et al. [15] Ragasa et al. [40] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
40 |
Campesterol |
Phoenix dactylifera, Mauritia flexuosa |
Whole plant extract Pulp extract |
Ahmed et al. [11] Pereira Freire et al. [22] |
|
|
41 |
Ursonic acid |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
42 |
Arborinol |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
43 |
3-acetyl ursolic acid |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
44 |
Fernenol |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
45 |
Arundoin |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
46 |
Cycloartenol |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
47 |
α-sitosterol |
Phoenix dactylifera |
Whole plant extract |
Ahmed et al. [11] |
|
|
48 |
β-sitosterol |
Mauritia flexuosa, Phoenix loureiroi, Phoenix paludosa, Salacca wallichiana |
Pulp extract Leaf extract Leaf extract Fruit extract |
Pereira Freire et al. [22] Mondal et al. [15] Alam et al. [41] Ragasa et al. [40] |
|
|
49 |
stigmastan-3,5-diene |
Mauritia flexuosa |
Pulp extract |
Pereira Freire et al. [22] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
50 |
β-Sitosterol 3-O-β-D-glucoside |
Phoenix dactylifera |
Leaf extract
|
Suleiman et al. [42] |
|
|
51 |
Ergost-4-en-3-one |
Phoenix paludosa |
Leaf extract |
Alam et al. [41] |
|
|
52 |
Ergost-4-ene-3,6-dione |
Phoenix paludosa |
Leaf extract |
Alam et al. [41] |
|
|
53 |
Stigmasta-4,22-diene-3,6-dione |
Phoenix paludosa |
Leaf extract |
Alam et al. [41] |
|
|
Vitamins |
|||||
|
54 |
Ascorbic acid (Vitamin C) |
Cocos nucifera, Mauritia flexuosa |
Liquid albumen extract Pulp extract |
Lima et al. [16] Pereira Freire et al. [22] |
|
|
55 |
Vitamin A |
Phoenix dactylifera |
Whole plant extract |
Ahmed et al. [11] |
|
|
56 |
Vitamin B1 |
Phoenix dactylifera |
Whole plant extract |
Ahmed et al. [11] |
|
|
57 |
Riboflavin (Vitamin B2) |
Phoenix dactylifera, Cocos nucifera |
Whole plant extract Liquid albumen extract |
Ahmed et al. [11] Lima et al. [16] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
58 |
Nicotinic acid (vitamin B3) |
Cocos nucifera |
Liquid albumen extract |
Lima et al. [16] |
|
|
59 |
Pantothenic Acid (vitamin B5) |
Cocos nucifera |
Liquid albumen extract |
Lima et al. [16] |
|
|
60 |
Vitamin E |
Mauritia flexuosa, Livistona australis |
Pulp extract Pulp extract |
Pereira Freire et al. [22] Kassem et al. [39] |
|
|
Carotenoids |
|||||
|
61 |
α-carotene |
Mauritia flexuosa |
Fruit extract |
Pereira Freire et al. [22] |
|
|
62 |
β-carotene |
Mauritia flexuosa |
Fruit extract |
Pereira Freire et al. [22] |
|
|
63 |
Lutein |
Mauritia flexuosa |
Fruit extract |
Pereira Freire et al. [22] |
|
|
Tocopherols |
|||||
|
64 |
α-tocopherol |
Mauritia flexuosa |
Pulp extract |
Pereira Freire et al. [22] |
|
|
65 |
β-tocopherol |
Mauritia flexuosa |
Pulp extract |
Pereira Freire et al. [22] |
|
|
66 |
δ-tocopherol |
Mauritia flexuosa |
Pulp extract |
Pereira Freire et al. [22] |
|
|
67 |
γ -tocopherol |
Mauritia flexuosa |
Pulp extract |
Pereira Freire et al. [22] |
|
|
Phenolic Compounds |
|||||
|
68 |
Quinic acid |
Mauritia flexuosa |
Pulp extract |
Pereira Freire et al. [22] |
|
|
69 |
Veratric acid |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
70
|
Rosmarinic acid |
Salacca edulis, |
Peel extract |
Kanlayavattanakul et al. [28] |
|
|
71 |
Caffeic acid |
Mauritia flexuosa, Ravenea rivularis, Salacca edulis, Hyphaene thebaica |
Pulp extract Leaf extract Peel extract Fruit extract |
Nonato et al. [24] da Silva et al. [21] Kanlayavattanakul et al. [28] Hussein et al. [29] |
|
|
72 |
Chlorogenic acid |
Mauritia flexuosa, Salacca edulis, Ravenea rivularis, Salacca zalacca |
Pulp extract Pulp extract Leaf extract Pulp extract |
Pereira Freire et al. [22] Kanlayavattanakul et al. [28] da Silva et al. [21] Saleh et al. [10] |
|
|
73 |
Syringic acid |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
74 |
Protocatechuic acid (3,4-dihydroxybenzoic acid) |
Cocos nucifera , Hyphaene thebaica, Livistona chinensis, Serenoa repens, Mauritia flexuosa |
Endocarp extract Fruit extract Fruit extract Pericarp extract Pulp extract |
Elsbaey et al. [43] Hussein et al. [29] Zeng et al. [44] Olennikov et al. [26] Pereira Freire et al. [22] |
|
|
75 |
p-Coumaric acid |
Serenoa repens, Livistona chinensis, Mauritia flexuosa |
Pericarp extract Fruit extract Pulp extract |
Olennikov et al. [26] Zeng et al. [44] Pereira Freire et al. [22] |
|
|
76 |
O-Coumaric acid |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
77 |
Ferulic acid |
Mauritia flexuosa, Ravenea rivularis, Areca catechu, Hyphaene thebaica, Serenoa repens, Salacca edulis |
Pulp extract Leaf extract Whole plant extract Fruit extract Pericarp extract Peel extract |
Pereira Freire et al. [22] da Silva et al. [21] Salehi et al. [30] Hussein et al. [29] Olennikov et al. [26] Kanlayavattanakul et al. [28] |
|
|
78 |
4-hydroxybenzaldehyde |
Livistona chinensis, Serenoa repens |
Fruit extract Pericarp extract |
Zeng et al. [44] Olennikov et al. [26] |
|
|
79 |
3-hydroxybenzoic acid |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
80 |
3-methoxybenzoic (m-anisic) |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
81 |
4-methoxybenzoic (p-anisic) |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
82 |
2,4-dihydroxybenzoic |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
83 |
2,5-dihydroxybenzoic |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
84 |
2,6-dihydroxybenzoic |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
85 |
3,5-dihydroxybenzoic |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
86 |
2-hydroxy-3-methoxybenzoic |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
87 |
2-hydroxy-5-methoxybenzoic |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
88 |
Isovanillic acid (3-hydroxy-4-methoxybenzoic acid) |
Livistona chinensis, Serenoa repens |
Fruit extract Pericarp extract
|
Zeng et al. [44] Olennikov et al. [26] |
|
|
89 |
4-hydroxybenzoic acid (p-hydroxybenzoic acid) |
Livistona chinensis, Serenoa repens |
Fruit extract Pericarp extract |
Zeng et al. [44] Olennikov et al. [26] |
|
|
90 |
Vanillic acid (4-hydroxy - 3- methoxybenzoic acid) |
Livistona chinensis, Serenoa repens, Areca catechu, Hyphaene thebaica |
Fruit extract Pericarp extract Whole plant extract Fruit extract |
Zeng et al. [44] Olennikov et al. [26] Salehi et al. [30] Hussein et al. [29] |
|
|
91 |
Isovanillin (3-hydroxy-4-methoxybenzaldehyde) |
Livistona chinensis, |
Fruit extract |
Zeng et al. [44] |
|
|
92 |
Gallic acid |
Hyophorbe indica, Salacca edulis, Hyphaene thebaica, Washingtonia robusta, Serenoa repens |
Leaf extract Peel extract Leaf extract Leaf extract Pericarp extract |
da Silva et al. [21] Kanlayavattanakul et al. [28] Eldahshan et al. [46] Selim et al. [38] Olennikov et al. [26] |
|
|
93 |
Resveratrol |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
94 |
Cinnamic acid |
Hyphaene thebaica |
Fruit extract |
Hussein et al. [29] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
95 |
3-O-Caffeoylshikimic acid |
Phoenix paludosa, Livistona chinensis |
Leaf extract Fruit extract |
Alam et al. [41] Zeng et al. [44] |
|
|
96 |
4-O-Caffeoylshikimic acid |
Phoenix paludosa |
Leaf extract |
Alam et al. [41] |
|
|
97 |
5-O-caffeoylshikimic acid |
Livistona chinensis |
Fruit extract |
Zeng et al. [44] |
|
|
98 |
1-p-Hydroxybenzoyl glycerol |
Brahea armata |
Fruit extract |
Hussein et al. [36] |
|
|
Amino Acid |
|||||
|
99 |
L-arginine |
Cocos nucifera |
Liquid albumen/solid albumen extract |
Lima et al. [16] |
|
|
Terpenoids |
|||||
|
1: Triterpenoid |
|||||
|
100 |
Betulinic acid |
Ravenea rivularis |
Leaf extract |
Mohammed and Fouad [31] |
|
|
101 |
Oleanolic acid |
Phoenix dactylifera |
Leaf extract |
Suleiman et al. [42] |
|
|
102 |
Lupeol |
Phoenix paludosa |
Leaf extract |
Alam et al. [41] |
|
|
103 |
Epilupeol |
Phoenix paludosa |
Leaf extract |
Alam et al. [41] |
|
|
104 |
Lupeol acetate |
Ravenea rivularis |
Leaf extract |
Mohammed and Fouad [31] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
Tannins |
|||||
|
105 |
Catechins |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
106 |
Epicatechins |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
Stilbenoid Derivatives |
|||||
|
107 |
Cassigarol G |
Cocos nucifera |
Endocarp extract |
Elsbaey et al. [43] |
|
|
108 |
Maackin A |
Cocos nucifera |
Endocarp extract |
Elsbaey et al. [43] |
|
|
109 |
Piceatannol |
Cocos nucifera, Aiphanes aculeata |
Endocarp extract Seed extract |
Elsbaey et al. [43] Lee et al. [47] |
|
|
110 |
Aiphanol |
Aiphanes aculeate |
Seed extract |
Lee et al. [47] |
|
|
111 |
Isorhapontigenin |
Aiphanes aculeate |
Seed extract |
Lee et al. [47] |
|
|
112 |
1-(3,5-Dihydroxyphenyl)-2-(3,4,5-trihydroxyphenyl) ethylene; (Z)-form, 4-methyl ether. (syn: (Z) 3,5,3',5'-Tetrahydroxy-4-methoxystilbene) |
Phoenix dactylifera
|
Stem extract
|
Mohammed and Fouad [31] |
|
|
113 |
1-(3,5-Dihydroxyphenyl)-2-(3,4,5-trihydroxyphenyl) ethylene; (E)-form, 4-methyl ether (sy.: (E) 3,5,3',5'-Tetrahydroxy-4-methoxystilbene) |
Phoenix dactylifera
|
Stem extract |
Mohammed and Fouad [31] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
Glyceryl Derivatives |
|||||
|
1: Monoacylglycerol |
|||||
|
114 |
1-Hexadecanoyl-sn-glycerol |
Livistona chinensis |
Root extract |
Zeng et al. [45] |
|
|
Alkaloids |
|||||
|
115 |
Arecoline |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
116 |
Arecaidine |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
117 |
guavacoline |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
118 |
guavacine |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
119 |
Isoguvacine |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
120 |
arecolidine |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
121 |
methyl nicotinate |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
122 |
Ethyl nicotinate |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
123 |
Nicotine |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
124 |
homoarecoline |
Areca catechu |
Whole plant extract |
Salehi et al. [30] |
|
|
Lignan Derivatives |
|||||
|
125 |
Quiquelignan A |
Calamus quiquesetinervius |
Stem extract |
Chang et al. [48] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
126 |
Quiquelignan B |
Calamus quiquesetinervius |
Stem extract |
Chang et al. [48] |
|
|
127 |
Quiquelignan C |
Calamus quiquesetinervius |
Stem extract |
Chang et al. [48] |
|
|
128 |
Quiquelignan D |
Calamus quiquesetinervius |
Stem extract |
Chang et al. [48] |
|
|
129 |
Quiquelignan E |
Calamus quiquesetinervius |
Stem extract |
Chang et al. [48] |
|
|
130 |
Quiquelignan F |
Calamus quiquesetinervius |
Stem extract |
Chang et al. [48] |
|
|
131 |
Quiquelignan G |
Calamus quiquesetinervius |
Stem extract |
Chang et al. [48] |
|
|
132 |
Quiquelignan H |
Calamus quiquesetinervius |
Stem extract |
Chang et al. [48] |
|
Table 1: Continued
|
Structure No. |
Compound Name |
Sources |
Part used |
Reference |
Structures |
|
Ceramide Derivatives |
|||||
|
133 |
(2S,3S,4R,9Z)-2-[(2R)-2-Hydroxytricosanoylamino]-9-octadecene-1,3,4-triol |
Livistona chinensis |
Root extract |
Mohammed and Fouad [31] |
|
|
134 |
1-O-β-D-Glucopyranosyl-(2S,3S,4R,9Z)-2-[(2R)-2-hydroxydocosanoylamino]-9-octadecene-1,3,4-triol |
Livistona chinensis |
Root extract |
Mohammed and Fouad [31] |
|
|
Sugars |
|||||
|
135 |
Mannose |
Phoenix paludosa |
Leaf extract |
Alam et al. [41] |
|
|
136 |
Maltose |
Phoenix dactylifera |
Fruit extract |
Mohammed and Fouad [31] |
|
|
137 |
Sucrose |
Phoenix dactylifera |
Fruit extract |
Mohammed and Fouad [31] |
|
|
138 |
β-D-Glucopyranosyl-(1→2)-β-D-fructofuranosyl-(6→6)-α-D-glucopyranoside |
Phoenix dactylifera |
Fruit extract |
Mohammed and Fouad [31] |
|
|
139 |
Maltotriose |
Phoenix dactylifera |
Fruit extract |
Mohammed and Fouad [31] |
|
|
Simple Phenolic Glycosides |
|||||
|
140 |
6'-O-(4-Hydroxybenzoyl)-β-glucose |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
|
141 |
6'-O-(3,4-Dihydroxybenzoyl)-β-glucose |
Serenoa repens |
Pericarp extract |
Olennikov et al. [26] |
|
This mentioned data revealed the chemotaxonomic classification of phytochemicals present in different genra of the family Arecaceae.
Biological Properties
The family Arecaceae has medicinal importance due to the presence of secondary metabolites such as glycosides, terpenoids, alkaloids, sterols, tannins, flavonoids, carotenoids and phenolic compounds. The biological activities of various species of the family Arecaceae are highlighted below in Table 2.
Antimicrobial Activity
In Vitro as well as In Vivo studies have shown that the leaf extract of Elaeis guineensis exhibits excellent antimicrobial activity, effective against both bacterial and fungal infections, especially against the yeast Candida albicans [49]. M. flexuosa induce susceptibility to conventional antibiotics in Gram-positive, Gram-negative and Candida species [24]. The most efficient extract against bacteria was methanolic extract of A. catechu (18Escherichia coli growth was prevented by a leaf extract from C. nucifera [52]. With minimal inhibitory concentrations ranging from 190 to 3120 g/mL, the extract of Syagrus coronata exhibited bactericidal activity [53].
The PKnB kinase enzyme is inhibited by extract of onecapaus bataua but bacterial growth is unaffected. It has an IC50 of 60.9 g/mL [54]. Staphylococcus aureus' growth was entirely prevented by the extract of Attalea speciosa at its highest dose (500 mg/mL) [55]. Gram-positive and Gram-negative bacteria were moderately sensitive to the aqueous methanol extract and ethyl acetate fraction of D. leptocheilos leaves [56].
When 30 µL of aqueous fruit extract of Borassus flabellifer was used, a 23mm-diameter zone of inhibition was seen against S. aureus [57]. Both S. aureus and E. coli were moderately susceptible to the mild bactericidal effects of aqueous fractions of C. mitis [58]. E. coli growth was slowed down by a S. edulis extract. At 100% concentration, the maximum inhibition zone was found (average diameter 18.783 mm) [59]. Bacillus subtillus, Escherichia coli, Pseudomonas aeruginosa and Candida albicans were all susceptible to the antibacterial effects of aqueous methanol extract of Hyophorbe verschaffeltii [60].
The PKnB kinase enzyme is inhibited by Euterpe precatoria but bacterial growth is unaffected. It has an IC50 of 77.4 g/mL [54]. When mixed with chloramphenicol, kanamycin, streptomycin, erythromycin and tetracycline, Raphia hookeri extract was able to reduce the antibiotic concentration by up to 50% [61]. Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa are all susceptible to the antibacterial effects of oil of Chamaerops humilis [55]. Strong antibacterial activity was demonstrated by the ethanolic extract of H. thebaica fruits against Salmonella typhi and Staphylococcus aureus [62]. Antibiotic-resistant bacteria strains were able to be inhibited and killed by the oil from Syagrus coronate [63]. Staphylococcus aureus, Escherichia coli, Pseudomonas aerigunosa and Bacillus cereus were all susceptible to methanolic extracts of leaves of Phoenix dactylifera. Gram-positive bacteria showed to be more susceptible than gram-negative bacteria [64].
Anti-Parasitic Activity
Hamsters infected with L. braziliensis were examined for response to ethyl acetate extract of Cocos nucifera. After 14 days, skin lesions in infected hamsters were reduced by the ethanolic extract of Cocos nucifera (300 mg/kg, 0.2 mL) [35].
Hepatoprotective Activity
It has been demonstrated that the aqueous dates extract has hepatoprotective properties [65]. It has been demonstrated that methanol extract of leaves of Elaeis guineensis works well as a hepatoprotectant [65]. According to this study, an aqueous flesh extract of Phoenix dactylifera can reduce the CCl4-induced hepatotoxicity [11]. Aqueous extract of Areca catechu provided significantly more protection against liver injury in rats at 2000 mg/kg (67-85%) compared to 500 and 1000 mg/kg (18-33%) [66]. D-galactosamine-induced histological alterations in liver were significantly inhibited by an ethanolic extract of Dypsis lutescens. The extract enhanced liver operations [67]. Both as an aphrodisiac and a cure for male impotence, date palm pollen can be used to manage sexual dysfunction [65].
Antioxidant Activity
The serum increases of the liver function markers ALT and AST caused by CCl4 were greatly reduced and returned to normal (p<0.001 and p<0.05, respectively) following treatment with H. verschaffeltii leaf extract [32]. High radical scavenging activity is present in the hydro-alcoholic pulp extract of B. guineensis against DPPH [68]. Strong antioxidant activity was demonstrated by the D. leptocheilos aqueous methanol extract and ethyl acetate fraction, with SC50 values of 12.8±0.56 µg/mL and 17±0.77 µg/mL, respectively [56]. The leaf hydro-ethanolic extract of Caryota urens has a significant antioxidant activity (21.25±4.51 mg/g) [57].
The antioxidant activity of extract of Borassus flabellifer was assessed every five minutes for the next thirty minutes. Thus, the DPPH percentage of radical scavenging was (56.69, 62.20, 62.99, 64.57, 66.14, 66.93 and 67.72%), correspondingly [57]. Ethyl acetate and butanol fractions of Rhapis excels demonstrated outstanding antioxidant activity in DPPH scavenging activity (86.2 and 75.6%, respectively) [69]. 107.6, 126.7, 172.7 and 196.3 g/mL were the IC50 values, respectively, for the Methanol/Ultrasonic (MU), Methanol/Water bath (MW), Ethanol/Ultrasonic (EU) and Ethanol/Water bath (EW) extracts of Hyphaene thebaica [62].
Fruit of L. australis can help prevent oxidative damage to physiologically active compounds and lipid peroxidation [39]. For DPPH radical scavenging, the IC50 values of methanolic extracts of endocarp and mesocarp of Calamus erectus are 0.10 and 0.12 mg/mL, respectively. When the concentration was raised, the antioxidant tests of both extracts improved [70]. When compared to aqueous extract (IC50 = 25 g/mL), methanolic extract of Dypsis lutescens had the highest level of radical scavenging activity (IC50 = 18 g/mL) [13]. At 100.0 μg/mL, all extracts of Euterpe precatoria demonstrated significant radical scavenging activity [71]. The inhibition rates of oxidation using 0.4 mL of extracts from the pulp and kernel cake of S. romanzoffiana were 97.00, 0.43% and 95.13 0.7%, respectively [72].
E. guineensis leaf methanol extract shows strong hepatoprotective effects against paracetamol-induced liver injury in mice [73]. Dictyosperma album leaves have much higher antioxidant activity than Archinto-phoenix alexandrae leaves, with values of 243.51 and 129.40 g AECE/g (IC50 = 60 and 108.5 g/mL, respectively) [12]. Phoenix dactylifera has been shown to have antioxidant and anti-mutagenic effects [11]. The ethyl acetate fraction of Sabal palmetto demonstrated strong free radical scavenging, reducing, hydrogen peroxide, nitric oxide and lipid peroxidation inhibition abilities [74]. In comparison to its pulp, aqueous, methanolic and ethanolic extracts of peel of tucum-do-cerrado showed increased antioxidant activity [75].
Analgesic Activity
When compared to a control group, the ethanolic extract of Cocos nucifera significantly reduced writhing by 24, 34 and 52.4% at doses of 50, 100 and 150 mg/kg, respectively [16]. At doses of 200, 400 and 600 mg/kg, p.o., of extract of Phoenix loureiroi, the percentage inhibition of writhes is 33.34, 57.04 and 66.62%, respectively [15]. At a dosage of 400 mg/kg, all the tested samples had variable substantial analgesic efficacy. The percentage of inhibition was higher in the entire ethanolic extract and EtOAc fraction of Caryota mitis than in the other samples (96.1 and 92.85%, respectively) [76].
In a hot plate test, oral administration of extract of Phoenix sylvestris at dosages of 300 and 450 mg/kg at 120 min (6.60±0.28 and 8.88±0.55) significantly lengthened the latency period to thermal stimuli [77]. In a formalin-induced test, methanolic extract (500 mg/kg) of Calamus rotang resulted in 68.47% inhibition while indomethacin provided 70.72% inhibition [78]. For dosages of 50, 100, 200 and 400 mg/kg, b.wt., respectively, the leaf extract of Areca catechu showed greater antinociceptive action (55.8, 57.7, 86.5 and 88.5%) than the stem extract (30.8, 36.6, 40.9 and 59.6%) [79]. The amount of writhing caused by acetic acid decreased significantly at dosages of 150 and 300 mg/kg of ethanolic extract of Borassus flabellifer (30.67±2.84 and 19.33±1.56, respectively) [80].
Renal Protective Activity
In a Wistar rat model, treatment with 10% coconut water lowered crystal formation in renal tissue and decreased the amount of crystals in urine. Additionally, the extract protected the kidneys from oxidative stress buildup and renal function decline [81]. Aqueous extract of date flesh effectively reduced the increases in plasma creatinine and urea brought on by GM nephrotoxicity and mitigated proximal tubular damage [82].
Cardioprotective Activity
Cardiovascular markers (CK-MB and troponin-T) are decreased in the serum at doses of 50, 100 or 200 mg/ 100 g of Cocos nucifer [83].
Anti-Hyperlipidemic Activity
L. australis ether extract reduced blood cholesterol and triglycerides by 50 and 43.6%, respectively [39].
Anti-Hypertensive Activity
In Vivo experiments using methanolic extract (PSLME) and hydro-alcoholic extract of Phoenix sylvestris (PSLHAE) show antihypertensive and anti-diabetic effects without producing toxicity [84]. At a dosage of 25 mg/kg, extract of Cocos nucifera significantly reduced mean systolic blood pressure (from 185.3±4.7 to 145.6±6.1 mm Hg) in the deoxycorticosterone acetate salt-induced hypertension model [16].
Anti-Platelet Activity
Adenosine diphosphate (ADP) and Ca+2 ionophore-induced aggregation were most effectively inhibited by A. catechu [85].
Anti-Trichomonal Activity
C. nucifera husk fibre crude methanol extract showed significant antitrichomonal action (IC50 value of 5.8 mg/mL) and activity was lower than that of metronidazole [16].
Effects on Bone Structure
Administration of virgin coconut oil significantly raised bone volume, avoided a decline in trabecular number and decreased trabecular separation [16].
Anti-Diabetic Activity
According to this investigation, the methanolic extract of Phoenix sylvestris inhibited both enzymes to varying degrees (26.45-78.48% for α-amylase and 38.28-76.07% for α-glucosidase) [86]. At 400 g/mL, ethanolic and methanolic extracts of Phoenix roebelenii showed the strongest inhibitory efficacy. The maximum inhibitory effect for both α-amylase (75.5±0.66%) and α-glucosidase (77.5±1.07%) was displayed by the ethanolic extract, whereas the activity of the methanolic extract was 70.4±0.62% for α-amylase and 75.5±0.09% for α-glucosidase [87]. The extract of Phoenix dactylifera reduced the levels of creatinine (0.95±0.1, 0.92±0.5 and 0.86±0.4 mg/dL), urea (52.33±0.1, 45.9±1.4 and 36.54±1.3 mg/dL) and alkaline phosphatase (212.39±3.2, 191.11±1.9 and 182.91±2.3 mg/dL), respectively, in the blood at doses of 150, 300 and 600 mg/kg [88].
The percentage of α-amylase enzyme inhibition was 8.42±0.97% for concentrated C. urens flour up to 5 mg/mL, compared to 10.77±2.64% for cooked flour [89]. Methanolic extracts of endocarp and mesocarp of Calamus erectus showed concentration-dependent inhibition of α-glucosidase (IC50 = 1.69 and 2.00 mg/mL) and α-amylase (IC50 = 2.74 and 3.30 mg/mL), respectively [70]. A 0.2 g/kg oral dose of the extract of Raphia gentiliana decreased blood sugar levels [90].
Anti-Cancer Activity
Although the components of dates have demonstrated an anticancer effect, their precise mode of action in the prevention of tumours is unknown [11]. Methanolic extracts of A. catechu have anticancer effects on breast cancer and oral squamous carcinoma cell lines (HSC-2) [13]. Isovitexin, a component of Dypsis lutescens, has anti-cancer properties [91]. Salacca fruit extract was cytotoxic to vero cells and a normal human fibroblast cell line [59].
The methanol extract of Elaeis guineensis significantly damaged MCF-7 cells in a dose-dependent manner [50]. Colorectal cancer Her2/CT26 cell proliferation was suppressed by trans-scirpusin A compound of Borassus flabellifer [92]. M. argun seeds were recommended for patients with human colon cancer risk factors and as an adjuvant therapy to cancer chemotherapy because they had chemopreventive effects against hepatocarcinogenesis [93].
Anti-Mutagenic Activity
Phoenix dactylifera fruit extract was found to have strong anti-mutagenic properties [11].
Diuretic Activity
At a dosage of 200 mg/kg, ethanol extract of Borassus flabellifer has significantly raised the urine levels of Na+, K+ and Cl [94].
Anti-Diarrheal Activity
Phoenix dactylifera aqueous extract can be used to treat diarrhoea by lowering the mean number of faeces compared to the saline control group [11].
Anti-Viral Activity
Extracts of Phoenix dactylifera exhibit a potent capacity to reduce infectivity of Pseudomonas phage ATCC 14209-B1 [95].
Anti-Fungal Activity
Phoenix dactylifera possesses antifungal qualities in both its leaves and its pits [11]. At a dosage of 100 g/mL, methanol extract of leaf of Borassus flabellifer demonstrated antifungal activity against a number of different fungal species, including Aspergillus flavus, Aspergillus fumigatus, Aspergillus niger, Candida albicans and Candida blanki [96].
Anti-Pyretic Activity
The ethanol extract of Phoenix loureiroi significantly reduced pyrexia in a dose-dependent manner [15]. Significant antipyretic efficacy was shown by n-hexane and aqueous fractions of Caryota mitis against yeast-induced hyperthermia [76]. Hyperthermia was significantly reversed by a Borassus flabellifer extract [80].
Delivery and Labor Relaxation Activity
Date fruit components have a substantial impact on pain relief and relaxation during labour [11].
Male Infertility and Testicular Dysfunction Activity
Phoenix dactylifera pollens can raise testosterone levels in cirrhotic patients, improving their sexual well-being [11].
Female Infertilitily and Harmone Levels Activity
The herb Phoenix dactylifera can also be used to treat female infertility brought on by hormonal imbalance [11].
Anti-Acetylcholinesterase (AChE) Activity
In Vitro spectrophotometric analysis was used to determine the anti-AChE activity. The enzyme was nearly completely inhibited by the extract of Areca catechu (90.1±0.4), demonstrating its high AChE inhibitory action [85].
Anti-Inflammatory Activity
C. nucifera root extracts reduced activities of 15-LOX (IC50 = 24.57±1.16 and IC50 = 8.31±0.73), sPLA2 (not determind (nd) and 24.68±0.08), COX-1 (nd) and 27.21±1.66) and COX-2 (nd and 39.41±1.36), respectively [97]. The microencapsulated oil from Acrocomia aculeata fruits also presented anti-inflammatory effects in paw edema and pleural edema models, both induced by carrageenan [18]. Methanolic extract of Ravenea rivularis at a dose of 125 g/mL showed an anti-inflammatory effect (66% inhibition) in comparison with dexamethasone at a dose of 50 ng/mL (65% inhibition) [32]. Aqueous extract of Areca catechu inhibited 5-LOX with an IC50 of 25.07 [13]. Mice's intestinal inflammation could be decreased by P. loureiroi extract when given orally at a dose of 5 mg/kg [98]. In Vivo tests on paw edoema and ear erythema brought on by croton oil revealed anti-inflammatory efficacy in oil derived from Euterpe oleracea fruits. 1226.8 mg/kg was the typical effective dose [99].
The activity of butyrylcholinesterase was reduced by B. odorata extract, suggesting that it may have anti-inflammatory effects [100]. At 800 µg, a root extract from Borassus flabellifer reduced hemolysis and denaturation by 50% [101]. Ethanolic extract of Dypsis lutescens shows the ability to inhibit the pro-inflammatory enzymes hyaluronic acid (37.78±1.26 and 29.79±1.27) and matrix metallopeptidase (814.51±20.25 and 629.18± 18.64), respectively, at two doses (250 and 500 mg/kg, p.o.) [67]. Anti-inflammatory potential of Caryota mitis, its extract was able to attenuate the inflammatory response in mice paw edema induced by carrageenan at a concentration of 400 mg/kg [102]. The methanolic extract (500 mg/kg) of Hyophorbe verschaffeltii demonstrated continuous and significant suppression of edoema by 48.54 and 44.2% at 8th and 12th h, respectively, demonstrating longer-lasting anti-inflammatory efficacy than diclofenac sodium (100 mg/kg) [60] (Figure 3).
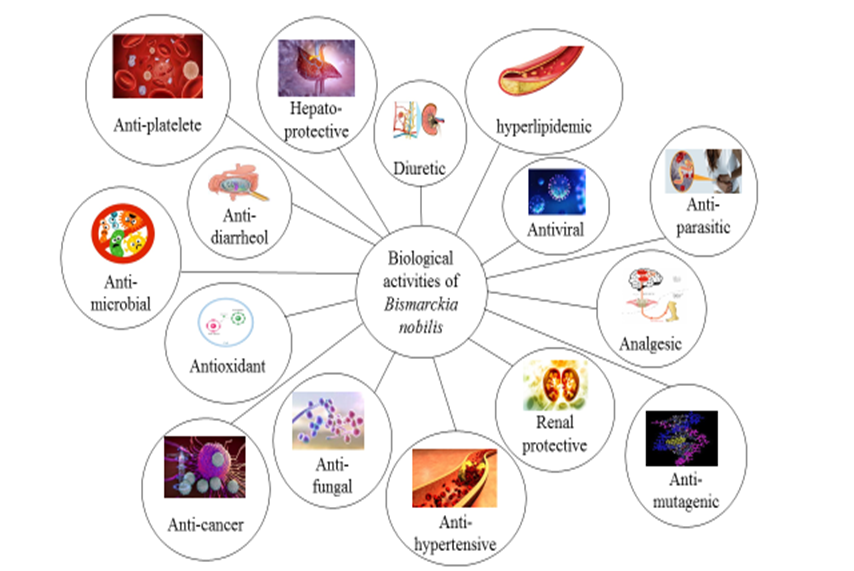
Figure 3: A Schematic Diagram of Biological Activities of Bismarckia nobilis
CONCLUSIONS
The species of Arecaceae family are widely distributed in Indonesia, Malaysia and Thailand. This review summarizes the biological activities, phytochemical studies and traditional uses of species of Arecaceae family. A total of 141 compounds are mentioned in this review; these secondary metabolites are very effective for the treatment of several diseases, such as hypertension, diabetes, spasms, nephritis, rheumatism, cough and digestive issues. Species of this family shows biological activities like hepatoprotective, anti-diabetic, anti-oxidant, antihypertensive, renal protective, cardioprotective, anti-microbial, anti-inflammatory, analgesic and anti-cancer. On the basis of data collected in this review, it is evident that Arecaceae family comprises a wide range of biologically important plants.
Future Perspective
According to this review, the Arecaceae family contains a number of species that need additional study in terms of characterization and phytochemical studies. We hope that information from this review will facilitate future research initiates to develop new medicinal plant-based medication for treating different diseases such as hypertension, nephritis, rheumatism, diabetes and cancer. It is necessary to conduct the clinical study to gain a better understanding of their safety and efficacy to ensure that it can be beneficial to the humanity.
Acknowledgement
The department of Pharmaceutical Chemistry, Faculty of pharmaceutical Sciences Govt. College University, Faisalabad supported this work.
REFERENCES
- Biswas, K. and R. Biswas. “DNA Molecular Markers Based Authentication of Herbal Drugs: A Review.” International Journal for Pharmaceutical Research Scholars, vol. 3, 2014, pp. 581-593.
- Afrin, S. et al. “Phytochemical Screening and Evaluation of Analgesic and Thrombolytic Activity of the Crude Methanolic Extract of Calamus rotang L. Leaves (Arecaceae).” International Journal of Pharmaceutical Sciences and Research, vol. 11, 2020, pp. 4293-4300.
- Alamgeer et al. “Anti-Arthritic Activity of Aqueous-Methanolic Extract and Various Fractions of Berberis orthobotrys Bien ex Aitch.” BMC Complementary and Alternative Medicine, vol. 17, 2017, pp. 1-16.
- Joharchi, M.R. and M.S. Amiri. “Taxonomic Evaluation of Misidentification of Crude Herbal Drugs Marketed in Iran.” Avicenna Journal of Phytomedicine, vol. 2, 2012, pp. 105-112.
- Mosihuzzaman, M. “Herbal Medicine in Healthcare-An Overview.” Natural Product Communications, vol. 7, 2012, pp. 807-812.
- Akinnawo, O. et al. “Aqueous Fraction of Alstonia boonei de Wild Leaves Suppressed Inflammatory Responses in Carrageenan and Formaldehyde Induced Arthritic Rats.” Biomedicine & Pharmacotherapy, vol. 86, 2017, pp. 95-101.
- Elgindi, M. et al. “Phytochemical Investigation and Antioxidant Activity of Hyophorbe verschaffeltii (Arecaceae).” Journal of Pharmacognosy and Phytochemistry, vol. 5, 2016a, pp. 39-46.
- Mohammed, M. et al. “Chemical and Biological Review on Various Classes of Secondary Metabolites and Biological Activities of Arecaceae (2006-2021).” Journal of Advanced Biomedical and Pharmaceutical Sciences, vol. 5, 2022, pp. 113-150.
- Silva, R. et al. “Emerging Source of Bioactive Compounds from Arecaceae Family: A Systematic Review.” Research, Society and Development, vol. 10, 2021, pp. 1-17.
- Saleh, M.S. et al. “Salacca zalacca: A Short Review of the Palm Botany, Pharmacological Uses and Phytochemistry.” Asian Pacific Journal of Tropical Medicine, vol. 11, 2018, pp. 645-652.
- Ahmed, A. et al. “Phytochemical and Therapeutic Evaluation of Date (Phoenix dactylifera): A Review.” Journal of Pharmacy and Alternative Medicine, vol. 9, 2016, pp. 11-17.
- Afifi, N.I. et al. “Phytochemical Composition and Antioxidant Activity of Two Species Related to Family Arecaceae.” Pharmaceutical Sciences Asia, vol. 49, 2022, pp. 43-50.
- Almaatty, A.H.A. et al. “Medicinal Usage of Some Arecaceae Family Members with Potential Anticancer Effect.” Journal of Biotech Research, vol. 13, 2022, pp. 55-63.
- Benmehdi, H. et al. “Phytochemical Investigation of Leaves and Fruits Extracts of Chamaerops humilis L.” Journal of Materials and Environmental Science, vol. 3, 2012, pp. 320-337.
- Mondal, S. et al. “Detection of Secondary Metabolites Using HPTLC and GC-MS Analysis and Assessment of Pharmacological Activities of Phoenix loureiroi Kunth (Arecaceae) Ethanolic Leaves Extract in the Management of Pyrexia, Pain and Inflammation.” Discovery Phytomedicine, vol. 8, 2021, pp. 67-82.
- Lima, E. et al. “Cocos nucifera (L.) (Arecaceae): A Phytochemical and Pharmacological Review.” Brazilian Journal of Medical and Biological Research, vol. 48, 2015a, pp. 953-964.
- Kamis, A. et al. “Preliminary Biochemical and Haematological Effects of Aqueous Suspension of Pulp of Hyphaene thebaica (L.) Mart in Rats.” Biokemistri, vol. 13, 2003, pp. 1-7.
- Lescano, C.H. et al. “Diuretic and Anti-Inflammatory Activities of the Microencapsulated Acrocomia aculeata (Arecaceae) Oil on Wistar Rats.” Journal of Medicinal Food, vol. 18, 2015, pp. 656-662.
- Saxena, M. et al. “Phytochemistry of Medicinal Plants.” Journal of Pharmacognosy and Phytochemistry, vol. 1, 2013, pp. 168-182.
- Agidew, M.G. “Phytochemical Analysis of Some Selected Traditional Medicinal Plants in Ethiopia.” Bulletin of the National Research Centre, vol. 46, 2022, pp. 1-22.
- Da Silva, R.S. et al. “Emerging Source of Bioactive Compounds from Arecaceae Family: A Systematic Review.” Research, Society and Development, vol. 10, 2021, pp. 1-17.
- Pereira Freire, J.A. et al. “Phytochemistry Profile, Nutritional Properties and Pharmacological Activities of Mauritia flexuosa.” Journal of Food Science, vol. 81, 2016, pp. R2611-R2622.
- Salib, J.Y. et al. “Anti-Diabetic Properties of Flavonoid Compounds Isolated from Hyphaene thebaica Epicarp on Alloxan Induced Diabetic Rats.” Pharmacognosy Research, vol. 5, 2013, pp. 22-29.
- Nonato, C. et al. “Chemical Analysis and Evaluation of Antioxidant and Antimicrobial Activities of Fruit Fractions of Mauritia flexuosa L. f. (Arecaceae).” PeerJ, vol. 6, 2018.
- El-Akad, R.H. et al. “Comparative Metabolites Profiling of Caryota mitis and Caryota urens via UPLC/MS and Isolation of Two Novel In Silico Chemopreventive Flavonoids.” Journal of Food Biochemistry, vol. 45, 2021, pp. 1-14.
- Olennikov, D. et al. “Phenolic Compounds from Serenoa repens Fruit.” Chemistry of Natural Compounds, vol. 49, 2013, pp. 526-529.
- Kassem, M.E. et al. “A Sulphated Flavone Glycoside from Livistona australis and Its Antioxidant and Cytotoxic Activity.” Natural Product Research, vol. 26, 2012, pp. 1381-1387.
- Kanlayavattanakul, M. et al. “Salak Plum Peel Extract as a Safe and Efficient Antioxidant Appraisal for Cosmetics.” Bioscience, Biotechnology and Biochemistry, vol. 77, 2013, pp. 1068-1074.
- Hussein, S.A.A. et al. “Phenolic Profiling of Hyphaene thebaica by LC-ESI-Mass: Iron Nanoparticles Significance and Cytotoxic Activity.” Egyptian Journal of Chemistry, vol. 64, 2021, pp. 1679-1686.
- Salehi, B. et al. “Areca catechu-From Farm to Food and Biomedical Applications.” Phytotherapy Research, vol. 34, 2020, pp. 2140-2158.
- Mohammed, M.H.H. and M.A. Fouad. “Chemical and Biological Review on Various Classes of Secondary Metabolites and Biological Activities of Arecaceae (2006-2021).” Journal of Advanced Biomedical and Pharmaceutical Sciences, vol. 5, 2022, pp. 113-150.
- Elgindi, M.R. et al. “Phytochemical Investigation and Antioxidant Activity of Hyophorbe verschaffeltii (Arecaceae).” Journal of Pharmacognosy and Phytochemistry, vol. 5, 2016b, pp. 39-46.
- Fathy, H.M. et al. “Chemical Constituents of Date Palm (Phoenix dactylifera L.) Fruit-Free Bunches and Their Biological Activities.” Pharmaceutical Chemistry Journal, vol. 55, 2021, pp. 384-391.
- Nonato, C.D.F.A. et al. “Chemical Analysis and Evaluation of Antioxidant and Antimicrobial Activities of Fruit Fractions of Mauritia flexuosa L. f. (Arecaceae).” PeerJ, vol. 6, 2018b, pp. 1-21.
- Lima, E. et al. “Cocos nucifera (L.) (Arecaceae): A Phytochemical and Pharmacological Review.” Brazilian Journal of Medical and Biological Research, vol. 48, 2015b, pp. 953-964.
- Hussein, S.A. et al. “Phenolics from Extracts of Brahea armata with Inhibitory Effect against 5alpha-Reductase Type-II.” Pharmazie, vol. 61, 2006, pp. 1034-1037.
- Abbas, F.A. and A.M. Ateya. “Estradiol, Esteriol, Estrone and Novel Flavonoids from Date Palm Pollen.” Australian Journal of Basic and Applied Sciences, vol. 5, 2011, pp. 606-614.
- Selim, N.M. et al. “Impact of Washingtonia robusta Leaves on Gamma Irradiation-Induced Hepatotoxicity in Rats and Correlation with STING Pathway and Phenolic Composition.” Pharmaceuticals, vol. 13, 2020, pp. 1-24.
- Kassem, M. et al. “Chemical Composition of the Lipophilic Fraction of Livistona australis R. Br. Mart. (Arecaceae) Fruit Pulp and Evaluation of Its Antioxidant and Antihyperlipidemic Activities.” Journal of Natural Products, vol. 7, 2014, pp. 210-221.
- Ragasa, C.Y. et al. “Chemical Constituents of Salacca wallichiana Mart.” International Journal of Current Pharmaceutical Review and Research, vol. 7, 2016, pp. 186-189.
- Alam, F. et al. “Phytochemical and Biological Investigations of Phoenix paludosa Roxb.” Dhaka University Journal of Pharmaceutical Sciences, vol. 8, 2009, pp. 7-10.
- Suleiman, R.K. et al. “New Constituents from the Leaves of Date Palm (Phoenix dactylifera L.) of Saudi Origin.” Molecules, vol. 26, 2021, pp. 1-10.
- Elsbaey, M. et al. “Chemical Constituents from Coconut Waste and Their In Silico Evaluation as Potential Antiviral Agents against SARS-CoV-2.” South African Journal of Botany, vol. 141, 2021, pp. 278-289.
- Zeng, X. et al. “Bioactive Phenolics from the Fruits of Livistona chinensis.” Fitoterapia, vol. 83, 2012a, pp. 104-109.
- Zeng, X. et al. “Cytotoxic Ceramides and Glycerides from the Roots of Livistona chinensis.” Fitoterapia, vol. 83, 2012b, pp. 609-616.
- Eldahshan, O.A. et al. “Potential Superoxide Anion Radical Scavenging Activity of Doum Palm (Hyphaene thebaica L.) Leaves Extract.” Records of Natural Products, vol. 2, 2008, pp. 83-93.
- Lee, D. et al. “A Novel Cyclooxygenase-Inhibitory Stilbenolignan from the Seeds of Aiphanes aculeata.” Organic Letters, vol. 3, 2001, pp. 2169-2171.
- Chang, C.L. et al. “Quiquelignan A-H, Eight New Lignoids from the Rattan Palm Calamus quiquesetinervius.” Bioorganic & Medicinal Chemistry, vol. 18, 2010, pp. 518-525.
- Vijayarathna, S. et al. “The Antimicrobial Efficacy of Elaeis guineensis: Characterization, In Vitro and In Vivo Studies.” Molecules, vol. 17, 2012, pp. 4860-4877.
- Vijayarathna, S. and S. Sasidharan. “Cytotoxicity of Methanol Extracts of Elaeis guineensis on MCF-7 and Vero Cell Lines.” Asian Pacific Journal of Tropical Biomedicine, vol. 2, 2012, pp. 826-829.
- Negi, B.S. and B.P. Dave. “In Vitro Antimicrobial Activity of Acacia catechu and Its Phytochemical Analysis.” Indian Journal of Microbiology, vol. 50, 2010, pp. 369-374.
- Joy, A. et al. “Antibacterial Screening and Phytochemical Powder Isolated from Dorsal Side of Leaves of Cocos nucifera (Arecaceae).” Journal of Pharmaceutical Sciences and Research, vol. 11, 2019, pp. 2555-2557.
- Hughes, A.F.D.S. et al. “Antimicrobial Activity of Syagrus coronata (Martius) Beccari.” Brazilian Archives of Biology and Technology, vol. 56, 2013, pp. 269-274.
- Aranaga, C. et al. “Antimycobacterial and PKnB Inhibitory Activities of Venezuelan Medicinal Plants.” International Journal of Microbiology, 2020, pp. 1-7.
- Barroqueiro, E.S. et al. “Immunomodulatory and Antimicrobial Activity of Babassu Mesocarp Improves Survival in Lethal Sepsis.” Evidence-Based Complementary and Alternative Medicine, 2016, pp. 1-7.
- Ibrahim, H. et al. “Phytochemical Screening and Biological Evaluation of Dypsis leptocheilos Leaves Extract and Molecular Docking Study of the Isolated Compounds.” International Journal of Pharmacy and Pharmaceutical Sciences, vol. 12, 2020, pp. 106-113.
- Rani, V.P. et al. “Phytochemical, Antioxidant and Antibacterial Activity of Aqueous Extract of Borassus flabellifer (L.).” Themed Section: Science and Technology, vol. 4, 2018, pp. 405-411.
- Abdelhakim, I.A. et al. “Chemical Constituents and Antimicrobial Activity of the Leaves of Caryota mitis Lour. (Arecaceae).” Journal of Medicinal Plants, vol. 5, 2017, pp. 250-255.
- Ridho, A. et al. “Insights of Phytoconstituents and Pharmacology Activities of Salacca Plants.” Journal of Applied Pharmaceutical Science, vol. 9, 2019, pp. 120-124.
- Aly, S.H. et al. “Hyophorbe verschaffeltii DNA Profiling, Chemical Composition of the Lipophilic Fraction, Antimicrobial, Anti-Inflammatory and Cytotoxic Activities.” Research Journal of Pharmaceutical, Biological and Chemical Sciences, vol. 7, 2016, pp. 120-130.
- Nguenang, G.S. et al. “Tristemma hirtum and Five Other Cameroonian Edible Plants with Weak or No Antibacterial Effects Modulate the Activities of Antibiotics against Gram-Negative Multidrug-Resistant Phenotypes.” The Scientific World Journal, 2018, pp. 1-12.
- Aboshora, W. et al. “Effect of Extraction Method and Solvent Power on Polyphenol and Flavonoid Levels in Hyphaene thebaica L. Mart (Arecaceae) Fruit and Its Antioxidant and Antibacterial Activities.” Tropical Journal of Pharmaceutical Research, vol. 13, 2014, pp. 2057-2063.
- Cibele, M.A.D.S.B. et al. “Syagrus coronata Seed Oils Have Antimicrobial Action against Multidrug-Resistant Staphylococcus aureus.” Journal of Medicinal Plants Research, vol. 10, 2016, pp. 310-317.
- Laouini, S. et al. “Phytochemical Analysis, Antioxidant and Antimicrobial Activities of Leaves Extract of Date Palm Grown in Algeria.” Journal of Fundamental and Applied Sciences, vol. 4, 2012, pp. 142-154.
- Gruca, M. et al. “African Palm Ethno-Medicine.” Journal of Ethnopharmacology, vol. 165, 2015, pp. 227-237.
- Pithayanukul, P. et al. “Hepatoprotective Potential of Extracts from Seeds of Areca catechu and Nutgalls of Quercus infectoria.” Molecules, vol. 14, 2009, pp. 4987-5000.
- El-Ghonemy, M.M. et al. “Hepatoprotective Activity of Dypsis lutescens against D-galactosamine-Induced Hepatotoxicity in Rats and Its Phytoconstituents.” Asian Pacific Journal of Tropical Biomedicine, vol. 9, 2019, pp. 467-473.
- López, S. et al. “Cytoprotective Action against Oxidative Stress in Astrocytes and Neurons by Bactris guineensis (L.) HE Moore (Corozo) Fruit Extracts.” Food and Chemical Toxicology, vol. 109, 2017, pp. 1010-1017.
- Hassanein, H. et al. “Polyphenolic Constituents and Antimicrobial Activity of Rhapis excels (Arecaceae, Coryphoideae).” Research Journal of Pharmaceutical, Biological and Chemical Sciences, vol. 6, 2015, pp. 1714-1722.
- Ghosal, M. and P. Mandal. “In Vitro Antidiabetic and Antioxidant Activity of Calamus erectus Roxb. Fruit: A Wild Plant of Darjeeling Himalaya.” International Journal of Pharmacy and Biological Sciences, vol. 4, 2013, pp. 671-684.
- Galotta, A.L.Q. et al. “Antioxidant and Cytotoxic Activities of ‘Açaí’ (Euterpe precatoria Mart.).” Química Nova, vol. 31, 2008, pp. 1427-1430.
- De Lacerda Coriolano, D. et al. “Biological Potential of Products Obtained from Palm Trees of the Genus Syagrus.” Evidence-Based Complementary and Alternative Medicine, 2021.
- Sasidharan, S. et al. “Antioxidant and Hepatoprotective Activity of Methanolic Extracts of Elaeis guineensis Jacq Leaf.” Pharmacologyonline, vol. 3, 2009, pp. 84-90.
- Ibrahim, A.Y. et al. “Evaluation of the Antioxidant, Anti-Inflammatory and Antitumor Properties of Sabal Grown in Egypt.” Egyptian Pharmaceutical Journal, vol. 16, 2017, pp. 168-183.
- Rosa, F.R. et al. “Phytochemical Compounds and Antioxidant Capacity of Tucum-do-Cerrado (Bactris setosa Mart), Brazil’s Native Fruit.” Nutrients, vol. 8, 2016, pp. 1-17.
- Abd Elhakim, I.A.E.A. et al. “Botanical and Biological Study of the Leaves of Caryota mitis Lour. Family Arecaceae Cultivated in Egypt.” Bulletin of Pharmaceutical Sciences, vol. 40, 2017b, pp. 71-95.
- Shajib, M.S. et al. “Antinociceptive and Neuropharmacological Activities of Methanol Extract of Phoenix sylvestris Fruit Pulp.” Frontiers in Pharmacology, vol. 6, 2015, pp. 1-12.
- Ripa, F.A. et al. “CNS Depressant, Analgesic and Anti-Inflammatory Activities of Methanolic Seed Extract of Calamus rotang Linn. Fruits in Rat.” Journal of Pharmacognosy and Phytochemistry, vol. 3, 2015, pp. 121-125.
- Barman, M.R. et al. “Antinociceptive Activity of Methanol Extract of Areca catechu L. Stems and Leaves in Mice.” Advances in Natural and Applied Sciences, vol. 5, 2011, pp. 223-226.
- Paschapur, M.S. et al. “Evaluation of the Analgesic and Antipyretic Activities of Ethanolic Extract of Male Flowers (Inflorescences) of Borassus flabellifer L. (Arecaceae).” International Journal of Pharmacy and Pharmaceutical Sciences, vol. 1, 2009, pp. 98-106.
- Gandhi, M. et al. “Prophylactic Effect of Coconut Water (Cocos nucifera L.) on Ethylene Glycol Induced Nephrocalcinosis in Male Wistar Rat.” International Brazilian Journal of Urology, vol. 39, 2013, pp. 108-117.
- Al-Qarawi, A. et al. “Nephroprotective Action of Phoenix dactylifera in Gentamicin-Induced Nephrotoxicity.” Pharmaceutical Biology, vol. 46, 2008, pp. 227-230.
- Chikku, A.M. and T. Rajamohan. “Dietary Coconut Sprout Beneficially Modulates Cardiac Damage Induced by Isoproterenol in Rats.” Bangladesh Journal of Pharmacology, vol. 7, 2012, pp. 258-265.
- Jain, P.K. et al. “Evaluation of Anti-Diabetic and Antihypertensive Activity of Phoenix sylvestris (L.) Roxb Leaves Extract and Quantification of Biomarker Quercetin by HPTLC.” Phytomedicine Plus, vol. 1, 2021, pp. 1-14.
- Ghayur, M.N. et al. “Identification of Antiplatelet and Acetylcholinesterase Inhibitory Constituents in Betel Nut.” Journal of Chinese Integrative Medicine, vol. 9, 2011, pp. 619-625.
- Kannaian, U.P.N. et al. “Phytochemical Composition and Antioxidant Activity of Coconut Cotyledon.” Heliyon, vol. 6, 2020.
- Mahalingam, G. “The In Vitro Antidiabetic Activity of Phoenix roebelenii Leaf Extract.” International Journal of Green Pharmacy, vol. 11, 2017.
- Ayatollahi, S.A. et al. “Antidiabetic Activity of Date Seed Methanolic Extracts in Alloxan-Induced Diabetic Rats.” Pakistan Veterinary Journal, vol. 39, 2019.
- Wimalasiri, G. et al. “Antioxidant and Anti-Diabetic Properties of Caryota urens (Kithul) Flour.” Procedia Food Science, vol. 6, 2016, pp. 181-185.
- Mpiana, P. et al. “Anti-Hyperglycemic Activity of Raphia gentiliana De Wild. (Arecaceae).” European Journal of Medicinal Plants, vol. 3, 2013.
- Chiduruppa, M. et al. “A Detailed Review on Dypsis lutescens (Arecaceae).” World Journal of Pharmacy and Pharmaceutical Sciences, vol. 7, 2018, pp. 1644-1650.
- Hong, E.-H. et al. “Trans-Scirpusin A Showed Antitumor Effects via Autophagy Activation and Apoptosis Induction of Colorectal Cancer Cells.” Oncotarget, vol. 8, 2017.
- Abdel-Hamid, N.M. et al. “Possible Protective Potency of Argun Nut (Medemia argun-An Ancient Egyptian Palm) against Hepatocellular Carcinoma in Rats.” Nutrition and Cancer, vol. 74, 2022, pp. 527-538.
- Chinnaswamy, P. et al. “Diuretic Effect of the Ethanolic and Aqueous Extract of Seedling of Borassus flabellifer.” Asian Journal of Microbiology, Biotechnology and Environmental Sciences, vol. 11, 2009, pp. 313-317.
- Jassim, S.A. and M.A. Naji. “In Vitro Evaluation of the Antiviral Activity of an Extract of Date Palm (Phoenix dactylifera L.) Pits on a Pseudomonas Phage.” Evidence-Based Complementary and Alternative Medicine, vol. 7, 2010, pp. 57-62.
- Jamkhande, P.G. et al. “Biological Activities of Leaves of Ethnomedicinal Plant, Borassus flabellifer Linn. (Palmyra Palm): An Antibacterial, Antifungal and Antioxidant Evaluation.” Bulletin of Faculty of Pharmacy, Cairo University, vol. 54, 2016, pp. 59-66.
- Wendkouni, L.M.E.B.K. et al. “Phytochemical and Biological Investigations of Extracts from the Roots of Cocos nucifera L. (Arecaceae) and Carica papaya L. (Caricaceae), Two Plants Used in Traditional Medicine.” African Journal of Biochemistry Research, vol. 15, 2021, pp. 28-35.
- Murugan, R. et al. “Study of Intestinal Anti-Inflammatory Activity of Phoenix loureiroi Kunth (Arecaceae) Fruit.” Biomedicine & Pharmacotherapy, vol. 93, 2017, pp. 156-164.
- Favacho, H.A. et al. “Anti-Inflammatory and Antinociceptive Activities of Euterpe oleracea Mart., Arecaceae, Oil.” Revista Brasileira de Farmacognosia, vol. 21, 2011, pp. 105-114.
- Ramos, V.P. et al. “Hypolipidemic and Anti-Inflammatory Properties of Phenolic Rich Butia odorata Fruit Extract: Potential Involvement of Paraoxonase Activity.” Biomarkers, vol. 25, 2020, pp. 417-424.
- Natarajan, K. and R. Sangeetha. “Evaluation of Anti-Inflammatory Property of the Roots of Borassus flabellifer.” International Journal of Research in Pharmaceutical Sciences, vol. 11, 2020, pp. 331-334.
- Abd Elhakim, I.A.E.A. et al. “Botanical and Biological Study of the Leaves of Caryota mitis Lour. Family Arecaceae Cultivated in Egypt.” Bulletin of Pharmaceutical Sciences. Assiut, vol. 40, 2017a, pp. 71-95.

